The potential for using alpha-linolenic acid as a therapy for cardiovascular disease
- *Corresponding Author:
- Dr Grant N Pierce
St Boniface Hospital Research Centre, 351 Tache Avenue, Winnipeg, Manitoba R2H 2A6.
Telephone: 204-235-3206
Fax: 204-235-0793
E-mail: gpierce@sbrc.ca
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact support@pulsus.com
[ft_below_content] =>Keywords
Alpha linolenic acid; Cardiovascular disease; Flaxseed; Heart disease; Polyunsaturated fatty acids
Functional Foods for the Prevention and Treatment of Cardiovascular Disease
Many natural food products possess a significant source of health benefit beyond normal nutrition. In recent years, a field of study has emerged that focuses on elucidating the beneficial or deleterious effects that specific foods and diets may have on various disease processes. One area of investigation is the role that specific nutrients play in the prevention of chronic cardiovascular disease (CVD) [1]. Largescale animal and human trials continue to identify the central importance of how different food sources affect modifiable cardiac risk factors such as diabetes, obesity and inflammation [2,3], diseases that are now reaching epidemic proportions.
One example of a diet-based clinical study that has changed foodintake guidelines to benefit cardiovascular health is the Dietary Approaches to Stop Hypertension (DASH) trial [4]. The DASH trial compared a typical Western diet with a diet rich in fruits, vegetables, low-fat dairy products and low in saturated fats. After two weeks of intervention, systolic blood pressure decreased significantly by 5.5 mmHg compared with the control Western diet [4,5]. This study demonstrates how simple alterations to a typical Western diet may result in a direct, positive net effect on health and the reduction of cardiovascular risk factors. Diet may also have significant implications in both the costs and benefits of long-term human health. As a result, many natural products that contain myriad properties that may directly influence chronic disease processes have been identified. Natural products that have been proven to elicit positive health effects are termed either ‘nutraceuticals’ or ‘functional foods’. As defined by Health Canada, a nutraceutical is “a product isolated or purified from foods that is generally sold in medicinal forms not usually associated with food. A nutraceutical is demonstrated to have a physiological benefit or provide protection against chronic disease”, whereas a “functional food is similar in appearance to, or may be, a conventional food, is consumed as part of a usual diet, and is demonstrated to have physiological benefits and/or reduce the risk of chronic disease beyond basic nutritional functions” [6]. Several nutraceuticals and functional foods have been shown to improve cardiovascular health [7,8].
The Role of Fibre and Omega-3 Fatty Acids
A product that has been clearly implicated in the reduction of cardiovascular risk is dietary fibre. Fibre possesses lipid-lowering, blood pressure- lowering and antioxidant effects [9,10]. Fibre is ingested, but is a nonabsorbable substance. Fibre may be ingested in either a soluble or insoluble form. Soluble fibre absorbs water, gaining viscosity and becomes fermented in the distal colon, while insoluble fibre binds gut contents, preventing absorption and increasing fecal mass [11,12]. Overall, dietary fibre interferes with the absorption of gut contents and increases gastric emptying. For example, fibre exhibits a cholesterollowering effect via its action as a bile salt sequestrant [13]. Soluble fibre binds bile salts in the gut, preventing their re-entry into circulation and eventual hepatic reabsorption; these salts are then excreted. The subsequent decrease in circulating bile salts causes an increase in expression of the hepatic enzyme alpha-cholesterol hydroxylase that transforms cholesterol to cholic acid to replenish lost bile salts [10]. This process further decreases the amount of endogenous cholesterol available for packaging and its entry into the circulation as a lipoprotein.
Recent findings suggest that the fibre viscosity may, in fact, be more closely linked with the lipid-lowering effects of dietary fibre. Vuksan et al [14] demonstrated that the lipid-lowering action of dietary fibre was significantly increased when a highly viscous form of fibre was administered in conjunction with a typical North American diet. This cholesterol-lowering effect was significantly greater than two other treatment arms containing greater amounts but less viscous forms of dietary fibre [14]. Contemporary guidelines suggest the ingestion of 30 g to 40 g of dietary fibre daily for a positive cardiovascular effect to be observed. The best dietary sources of fibre include whole fruits, vegetables, seeds and pulse crop products such as beans [15].
An example of a seed that provides a significant source of fibre is flaxseed (Linum usitatissimum). Flaxseed is composed of three major components: seed, lignin and oil. The fibre components found within the seed and lignin have been attributed to the modest cholesterollowering effects observed with the ingestion of flaxseed [16]. In total, dietary fibre accounts for 28% of flaxseed by weight [17]. Flaxseed has been identified as a functional food, and has been implicated in the reduction of primary cardiovascular risk and possible secondary prevention of CVD. Beyond flaxseed’s fibre component, it is believed to be beneficial for cardiovascular health due to its high concentration of the omega-3 polyunsaturated fatty acid (PUFA) alpha-linolenic acid (ALA). Flaxseed is one of the richest dietary sources of ALA [18]. Approximately 50% of the oil component of flax is ALA.
Omega-3 Fatty Acid Structure and Metabolism
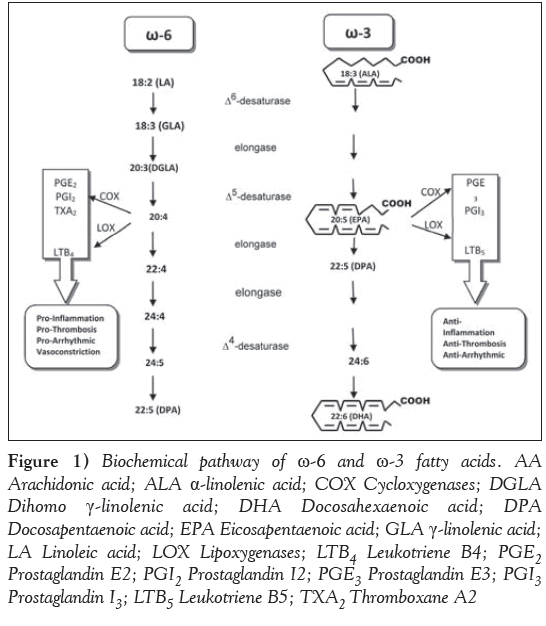
In terms of chemical structure, omega-3 fatty acids are long-chain fatty acids chemically configured in a cis conformation and contain two double bonds within the chain. The final double bond is located at the third-last carbon of the chain. Omega-3 fatty acids are complex PUFAs that require more energy to make bioavailable than various other food molecules such as carbohydrates or saturated fats. As a result, omega-3 fatty acids have a much slower rate of metabolism. Omega-3 fatty acids are structurally similar to their omega-6 fatty acid counterparts. Omega-6 fatty acids, however, are easily converted to arachadonic acid. From arachadonic acid, a series of proinflammatory downstream products are synthesized such as prostaglandins and thromboxanes (Figure 1). Omega-3 and omega-6 fatty acids are not interconverted within humans because of the lack of the proper desaturase enzyme. As a result, omega-3 and omega-6 fatty acids compete for enzymatic metabolism with other PUFAs and eventually eicosinoids, which may confer either pro- or anti-inflammatory downstream effects (Figure 1). Under normal circumstances, omega-3 fatty acids are actually preferentially bound by metabolizing enzymes.
Figure 1: Biochemical pathway of ω-6 and ω-3 fatty acids. AA
Arachidonic acid; ALA α-linolenic acid; COX Cycloxygenases; DGLA
Dihomo γ-linolenic acid; DHA Docosahexaenoic acid; DPA
Docosapentaenoic acid; EPA Eicosapentaenoic acid; GLA γ-linolenic acid;
LA Linoleic acid; LOX Lipoxygenases; LTB4 Leukotriene B4; PGE2 Prostaglandin E2; PGI2 Prostaglandin I2; PGE3 Prostaglandin E3; PGI3 Prostaglandin I3; LTB5 Leukotriene B5; TXA2 Thromboxane A2
While ancient diets provided an omega-6 to omega-3 ratio of approximately one, today’s guidelines suggest a ratio of 4:1 for maintenance of good cardiovascular health. Many estimates, however, place current ratios of omega-6 to omega-3 approaching 20:1 in a typical Western diet [19]. This provides the body with an exponentially larger amount of omega-6 fatty acids than omega-3s. As a result, the abundance of omega-6 fatty acids overcomes the preferential binding of omega-3 fatty acids by metabolizing enzymes. An increased amount of omega-6 fatty acids become bound and form myriad proinflammatory eicosanoid molecules. Over time, this process may significantly increase an individual’s overall inflammatory state which, in turn, has the potential to significantly accelerate chronic disease processes such as atherosclerosis. Omega-3 fatty acids actually provide an opposing effect when metabolized. While ALA possesses independent antiinflammatory effects [20-22], it may also be metabolized to other PUFAs such as eicosapentaenoic acid (EPA) and, to a much lesser extent, docosahexaenoic acid (DHA) (Figure 1). These PUFAs stimulate the synthesis of eicosanoids, which decrease platelet adhesion and thrombosis [23,24].
Thus, in contrast to omega-6 fatty acids, omega-3 fatty acids confer anti-inflammatory characteristics and have been shown to clinically lower circulating markers of inflammation such as C-reactive protein and serum amyloid A [25,26]. The ill-proportioned ratio of omega-6 to omega-3 may also be responsible for the negative or null results obtained in recent clinical trials investigating possible effects on CVD. In fact, the omega 6:omega 3 ratio is a possible source of error in the results of the recent Alpha Omega trial [27]. In this dietary study, a margarine supplement rich in various omega-3 fatty acids was administered to postmyocardial infarction (MI) patients undergoing modern optimal medical therapy [27]. No positive beneficial cardiovascular effects were observed; however, the omega-6:omega-3 ratio in the margarine supplement was very high – in fact, it was 2.3 times higher than the ratio utilized in the Lyon Heart Diet study [27]. In light of this fact, added to the possibility that the Alpha Omega trial may have been underpowered, the results may become less significant. It is quite possible that the high omega-6 component of the dietary intervention may have masked the cardioprotective effects of the low-dose omega-3 fatty acid supplementation. Many of the large clinical trials that demonstrated a potential benefit of omega-3 fatty acids for secondary prevention of CVD also achieved a concomitant lowering of the omega-6:omega-3 ratio [28,29]. Thus, the ability of omega-3 fatty acids to play a significant role in the secondary prevention of CVD remains a viable possibility.
Omega-3 Fatty Acids and CVD
Omega-3 fatty acids may be obtained from several dietary sources. The most common source is fish and marine products. Fish provide an excellent source of the long-chain omega-3 fatty acids EPA and DHA. In 1973, Bang et al [30] first reported the beneficial effects of a marine based diet on cardiovascular health [30]. EPA and DHA obtained from marine sources have been demonstrated to reduce the risk of MI, sudden cardiac death and all-cause mortality [31,32]. The positive effects on cardiovascular health from omega-3 fatty acids derived from fish has prompted investigators to explore the potential benefits of other omega-3 fatty acids. Omega-3 fatty acids such as the short-chain fatty acids derived from plant sources may elicit similar protective effects and serve as an available alternative for people with allergies or general intolerance to fish. Increasing concerns about the contamination of marine products with elevated levels of substances toxic to the humans, such as mercury (particularly in commercially concentrated fish oils), has made the identification of alternative sources of omega-3 fatty acids even more important.
One example of a plant source rich in an omega-3 fatty acid is flaxseed. Flaxseed is also called linseed. Canada is one of the largest producers of flaxseed. Flaxseed is unique in that it is one of the richest sources of ALA. ALA is another omega-3 PUFA, apart from EPA and DHA, which has been independently identified as possessing several cardioprotective properties. Flaxseed exhibits cholesterol-lowering, antiarrhythmic, antiatherogenic and blood pressure-lowering effects [18,33].
It is believed that many of these beneficial effects may be due to its high content of ALA. Many of these effects have been well-demonstrated in various species of animals, as well as in healthy individuals. Current research is expanding the benefits of ALA to patients with pre-existing cardiovascular disease. This research will help to define the role of ALA beyond primary prevention of CVD and elucidate the possibility of ALA therapy for secondary treatment of CVD. The FlaxPAD study is a recent example of a local, large-scale clinical trial designed to determine the health effects of long-term dietary supplementation with flaxseed in patients with peripheral artery disease [34].
Absorption Characteristics and Bioavailability of ALA
Essential fatty acids, such as ALA, must be ingested and absorbed because the body does not have the capacity to synthesize them. Given the benefits of increasing circulating ALA levels in blood and tissues, it is important to identify mechanisms that may influence the absorption and overall bioavailability of ALA.
Presently, there is limited information about the characteristics of ALA absorption. Our laboratory, however, has elucidated the dosage and form of flaxseed that provides the greatest bioavailability of ALA in humans. In one study [35], three types of flax-supplemented muffins, containing either whole or milled flaxseed, or flax oil and each providing 6 g of ALA were administered daily to a population 18 to 49 years of age over a 12-week period. It was determined that flax oil provided the greatest bioavailability of ALA over 12 weeks of supplementation compared with whole or milled flaxseed. Another study from our laboratory demonstrated a significant rise in plasma ALA concentrations following only four weeks of flax oil supplementation [36]. Milled flaxseed, however, provided a similar bioavailability of ALA and was deemed to be significantly more tolerable over the longer 12-week supplementation period [35]. Furthermore, milled flaxseed does not degrade as quickly as flax oil and appears to be more easily integrated into an individuals’s daily diet. The bioavailability of ALA obtained from ground flaxseed is also unaffected by a subject’s age. Plasma ALA concentrations remained similar when comparing a young adult (18 to 29 years of age) and older adult (45 to 69 years of age) human population [37].
It remains largely unclear what effect interactions with other substances within the gut or enterocytes may have on the absorption and metabolism of ALA and other omega-3 fatty acids. Supplementation of the diet with cholesterol has led to an approximately 120-fold increase in plasma ALA levels in animals [38,39]. The increase in intestinal cholesterol is believed to stimulate ALA absorption through the intestinal wall [40,41]. However, essential fatty acids are not absorbed directly with cholesterol. While sterol transport occurs via the NPC1L1 transporter in the intestinal brush border in the gut, fatty acids are primarily absorbed passively via fatty acid binding transport proteins along the intestinal brush border in the small intestine [42]. Under normal conditions, essential fatty acids have a very high rate of absorption, exceeding 90% [43]. A potential mechanism for the observed increase in fatty acid absorption in the presence of increased gut cholesterol may be based on an increase in micelle formation and overall affinity and/or interaction of the intestinal brush border and gut contents [40,41]. Thus, conversely, molecules that interfere with the formation of micelles or the affinity of gut contents for the intestinal brush border may markedly affect the overall bioavailability of cardioprotective fatty acids.
Animal Research Demonstrating the Cardioprotective Effects of ALA
In recent years, several animal studies have demonstrated several cardioprotective effects of ALA [18,24]. These have included effects on the vasculature as well as directly on the heart. With respect to the former effects, Bassett et al [18,44] demonstrated in an LDL receptor −/− mouse model that the ingestion of flaxseed significantly attenuated the development of atherosclerosis induced by a diet either rich in cholesterol or industrial trans-fats. Prasad [45] also demonstrated the ability of flaxseed to reduce the amount of atherosclerotic plaque formations in hypercholesterolemic rabbits by 46% when ingested over eight weeks despite no change in serum lipid levels over that time. Recently, Francis et al [46] demonstrated that flaxseed not only slowed the progression of atherosclerosis, but also induced its regression once the plaques were established.
Flaxseed has also been associated with improved vascular function. The ability of an artery to contract or relax is mediated, in part, via systemic or paracrine signalling of the endothelial vessel lining. Dupasquier et al [39] determined that following eight weeks of dietary supplementation with cholesterol and flax in rabbits, the presence of 10% flax in the diet significantly reduced the progression of atherosclerosis compared with 0.5% cholesterol supplementation alone. Even the combined treatment of a diet containing both 0.5% cholesterol and 10% flax demonstrated the same antiatherogenic effect. This observation, however, was attenuated at 16 weeks. Despite this, flaxsupplemented groups still elicited a normalizing effect on aortic relaxation fully at eight weeks and partially at 16 weeks. These findings suggest a protective effect on endothelial-dependent vessel relaxation by flaxseed supplementation in hypercholesterolemic conditions [39]. Talom et al [47] also demonstrated in hypertensive rats that a diet rich in flaxseed improved acetylcholine-mediated endothelial relaxation. Disturbances in vascular function may be caused by an insult to the endothelium at the beginning of the atherosclerosis or the exacerbation of pre-existing atherosclerotic disease. The ability to positively affect vascular function may be one mechanism responsible for the beneficial effects of flaxseed in the primary prevention of CVD.
The direct effects of flaxseed on cardiac function have also been investigated. Ander et al [48] reported that the ingestion of flaxseed by rabbits over a 16-week period elicited an antiarrhythmic effect in hearts during an in vitro ischemia-reperfusion challenge. The presence of flax in the diet caused a decrease in ventricular fibrillation and a shortening of the QT interval either alone or in conjunction with cholesterol supplementation. Similar anti-arrhythmic effects have been reported for different species and with other omega-3 fatty acidbased interventions [49,50]. ALA may achieve its positive effects on postischemic cardiac function through an inhibition of Na+/Ca2+ exchange [51]. It may also achieve its effects through an antioxidative mechanism. Oxygen-derived free radicals are believed to participate in ischemic injury to the heart [52-54]. However, while the lignan component of flaxseed (secoisolariciresinol diglucoside [SDG]) has marked antioxidative effects [55,56], SDG is not absorbed from a flaxseedsupplemented diet. It is metabolized in the gut to enterolignans that can then circulate in the blood. Thus, the ultimate clinical relevance of SDG therapy remains unclear. This finding has not been translated into positive clinical trials [18].
Clinical Trials Demonstrating the Cardioprotective Effects of ALA
A number of large-scale clinical trials have supported the cardiovascular benefits of a diet rich in omega-3 fatty acids [17,29,33,57]. Several studies have implicated the significant role ALA supplementation may play in the primary prevention of CVD. Ascherio et al [58] studied 43,757 men 40 to 75 years of age and free of previous CVD or diabetes. After six years of follow-up, it was determined that following a multivariate analysis, ALA was inversely correlated with coronary artery disease. Furthermore, this effect was not demonstrated by marine omega-3 fatty acids such as EPA. Thus, ALA may function by an independent cardioprotective mechanism other than via its conversion to long-chain fatty acids such as EPA or DHA [58].
In a prospective, 10-year study involving 76,283 women, Hu et al [59] demonstrated that a long-term ingestion of a diet rich in ALA provided a significant protective effect against fatal MI in women who had no previous ischemic heart disease [59]. Djoussé et al [60] investigated the relationship between ALA intake and coronary artery disease in 4584 male and female study participants. ALA intake was significantly and inversely correlated with the prevalence OR (95% CIs) of coronary artery disease. Those subjects (male or female) with the highest ALA intake had the greatest reduction in risk [60].
Another large, population-based, prospective study conducted by Albert et al [61] analyzed the ability of ALA to protect against sudden cardiac death in 76,763 women. After 18 years of follow-up, the investigators determined that the quintiles of women with the two highest daily intakes of ALA had a 38% and 40% reduction in sudden cardiac death, respectively [61].
There is a growing body of evidence supporting the role of omega-3 fatty acids for the secondary prevention of CVD. For example, the Diet and Reinfarction Trial (DART) [62] was one of the first large clinical trials to investigate the potential role that omega-3 fatty acids play in the secondary prevention of CVD. The DART trial enrolled >2000 men who had recently experienced MI. Subjects were randomly assigned to one of three dietary interventions for two years. One treatment arm ingested fatty fish two to three times per week. After two years of dietary intervention and consultation, this group had a 29% reduction in all-cause mortality. Although there was no significant change in the primary end point of re-infarction or ischemic heart death among any of the groups, the marked increase in survival in the fatty fish group lead to the conclusion that a diet supplemented with fatty fish that are rich in omega-3 fatty acids may decrease risk of mortality in men following a MI [62].
The GISSI-Prevenzione study [29] was a large-scale prospective clinical trial with >11,000 study participants. Again, this trial enrolled patients who had recently experienced their first MI. The study demonstrated that over 3.5 years of intervention, a significant 10% decrease in relative risk of the primary end point of death, nonfatal MI or stroke was observed in the treatment group receiving regular omega-3 fatty acid supplementation. There was also a small, but significant reduction in circulating triglyceride levels in the omega-3 fatty acids group [29].
In terms of ALA, the Lyon Diet Heart Study [28] examined the cardioprotective role that a Mediterranean-based diet rich in omega-3 fatty acids, specifically ALA, may exhibit on the secondary prevention of CVD. Patients were randomly assigned to either control or dietary intervention during their hospital stay following their first MI. The study investigated the recurrence of secondary events such as cardiac death, recurrent MI, unstable angina and heart failure over a 46-month period. The Lyon Diet Heart Study demonstrated a protective effect of approximately 15% against a second event in patients receiving the dietary intervention for the duration of the study. Furthermore, ALA was the only fatty acid to be significantly associated with cumulative survival and the absence of a second MI at follow-up [28,63].
Given the multitude of cardioprotective properties elicited by omega-3 fatty acids, and ALA specifically, it is increasingly important to understand the factors that influence its mechanisms of action. Recent trials involving hypertensive patients supplemented with flaxseed may provide some answers. In a recent, placebo-controlled, double-blinded trial in which patients with peripheral arterial disease were administered foods that contained 30 g of milled flaxseed over one year, significant decreases in both systolic (15 mmHg) and diastolic (7 mmHg) blood pressure were observed in hypertensive patients [64]. This antihypertensive effect was associated with plasma ALA levels in these patients [64] and has been recently attributed to changes in the fatty acid-derived circulating oxylipin profile [65]. Further work to understand more fully the characteristics and mechanism responsible for the antihypertensive actions of dietary flaxseed are warranted and important in view of the powerful relationship between hypertension and significant clinical cardiovascular events.
Acknowledgements
This work is funded by the CIHR and St Boniface Hospital Research Foundation.
References
- Moore LL. Functional foods and cardiovascular disease risk: Building the evidence base. Curr Opin Endocrinol Diabetes Obes 2011;18:332-5.
- Alissa EM, Ferns GA. Functional foods and nutraceuticals in the primary prevention of cardiovascular diseases. J Nutr Metab 2012;2012:569486.
- Alberti KG, Eckel RH, Grundy SM, et al. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009;120:1640-5.
- Appel LJ, Moore TJ, Obarzanek E, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N Engl J Med 1997;336:1117-24.
- Champagne CM. Dietary interventions on blood pressure: The Dietary Approaches to Stop Hypertension (DASH) trials. Nutr Rev 2006;64:S53-6.
- Health Canada. Managing Health Claims for Foods in Canada: Towards a Modernized Framework, 1998. <http://www.hc-sc.gc.ca/ fn-an/label-etiquet/claims-reclam/nutra-funct_foods-nutra-fonct_ aliment-eng.php> (Accessed July 14, 2014).
- Mozaffarian D. Fish and n-3 fatty acids for the prevention of fatal coronary heart disease and sudden cardiac death. Am J Clin Nutr 2008;87:1991S-6S.
- Liu S, Manson JE, Lee IM, et al. Fruit and vegetable intake and risk of cardiovascular disease: The Women’s Health Study. Am J Clin Nutr 2000;72:922-8.
- Keys A, Grande F, Anderson JT. Fiber and pectin in the diet and serum cholesterol concentration in man. Proc Soc Exp Biol Med 1961;106:555-8.
- Sanchez-Muniz FJ. Dietary fibre and cardiovascular health. Nutr Hosp 2012;27:31-45.
- Anderson JW, Baird P, Davis RH Jr, et al. Health benefits of dietary fiber. Nutr Rev 2009;67:188-205.
- Roberfroid M. Dietary fiber, inulin, and oligofructose: A review comparing their physiological effects. Crit Rev Food Sci Nutr 1993;33:103-48.
- Dietary fiber as a binder of bile salts. Nutr Rev 1977;35:183-5.
- Vuksan V, Jenkins AL, Rogovik AL, Fairgrieve CD, Jovanovski E, Leiter LA. Viscosity rather than quantity of dietary fibre predicts cholesterol-lowering effect in healthy individuals. Br J Nutr 2011;106:1349-52.
- Mann J. Dietary carbohydrate: Relationship to cardiovascular disease and disorders of carbohydrate metabolism. Eur J Clin Nutr 2007;61(Suppl 1):S100-11.
- Prasad K. Flaxseed and cardiovascular health. J Cardiovasc Pharmacol 2009;54:369-77.
- Bloedon LT, Balikai S, Chittams J, et al. Flaxseed and cardiovascular risk factors: Results from a double blind, randomized, controlled clinical trial. J Am Coll Nutr 2008;27:65-74.
- Bassett CM, Rodriguez-Leyva D, Pierce GN. Experimental and clinical research findings on the cardiovascular benefits of consuming flaxseed. Appl Physiol Nutr Metab 2009;34:965-74.
- Simopoulos AP. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother 2002;56:365-79.
- Yoneyama S, Miura K, Sasaki S, et al. Dietary intake of fatty acids and serum C-reactive protein in Japanese. J Epidemiol 2007;17:86-92.
- Poudel-Tandukar K, Nanri A, Matsushita Y, et al. Dietary intakes of alpha-linolenic and linoleic acids are inversely associated with serum C-reactive protein levels among Japanese men. Nutr Res 2009;29:363-70.
- Erdinest N, Shmueli O, Grossman Y, Ovadia H, Solomon A. Anti-inflammatory effects of alpha linolenic acid on human corneal epithelial cells. Invest Ophthalmol Vis Sci 2012;53:4396-406.
- Kinsella JE, Lokesh B, Stone RA. Dietary n-3 polyunsaturated fatty acids and amelioration of cardiovascular disease: Possible mechanisms. Am J Clin Nutr 1990;52:1-28.
- Bloedon LT, Szapary PO. Flaxseed and cardiovascular risk. Nutr Rev 2004;62:18-27.
- Bemelmans WJ, Lefrandt JD, Feskens EJ, et al. Increased alphalinolenic acid intake lowers C-reactive protein, but has no effect on markers of atherosclerosis. Eur J Clin Nutr 2004;58:1083-9.
- Rallidis LS, Paschos G, Liakos GK, Velissaridou AH, Anastasiadis G, Zampelas A. Dietary alpha-linolenic acid decreases C-reactive protein, serum amyloid A and interleukin-6 in dyslipidaemic patients. Atherosclerosis 2003;167:237-42.
- Rauch B, Schiele R, Schneider S, et al. OMEGA, a randomized, placebo-controlled trial to test the effect of highly purified omega-3 fatty acids on top of modern guideline-adjusted therapy after myocardial infarction. Circulation 2010;122:2152-9.
- de Lorgeril M SP, Martin JL, Monjaud I, Delaye J, Mamelle N. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon Diet Heart Study. Circulation 1999;99:779-85.
- Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSIPrevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto miocardico. Lancet 1999;354:447-55.
- Bang HO, Dyerberg J, Hjoorne N. The composition of food consumed by Greenland Eskimos. Acta Med Scand 1976;200:69-73.
- Trikalinos TA, Lee J, Moorthy D, et al. Effects of eicosapentaenoic acid and docosahexanoic acid on mortality across diverse settings: Systematic review and meta-analysis of randomized trials and prospective cohorts: Nutritional Research Series, Vol 4. Rockville (MD), 2012.
- Marik PE, Varon J. Omega-3 dietary supplements and the risk of cardiovascular events: A systematic review. Clin Cardiol 2009;32:365-72.
- Rodriguez-Leyva D, Dupasquier CM, McCullough R, Pierce GN. The cardiovascular effects of flaxseed and its omega-3 fatty acid, alpha-linolenic acid. Can J Cardiol 2010;26:489-96.
- Leyva DR, Zahradka P, Ramjiawan B, Guzman R, Aliani M, Pierce GN. The effect of dietary flaxseed on improving symptoms of cardiovascular disease in patients with peripheral artery disease: Rationale and design of the FLAX-PAD randomized controlled trial. Contemp Clin Trials 2011;32:724-30.
- Austria JA, Richard MN, Chahine MN, et al. Bioavailability of alpha-linolenic acid in subjects after ingestion of three different forms of flaxseed. J Am Coll Nutr 2008;27:214-21.
- Kaul N, Kreml R, Austria JA, et al. A comparison of fish oil, flaxseed oil and hempseed oil supplementation on selected parameters of cardiovascular health in healthy volunteers. J Am Coll Nutr 2008;27:51-8.
- Patenaude A, Rodriguez-Leyva D, Edel AL, et al. Bioavailability of alpha-linolenic acid from flaxseed diets as a function of the age of the subject. Eur J Clin Nutr 2009;63:1123-9.
- Dupasquier CM, Dibrov E, Kneesh AL, et al. Dietary flaxseed inhibits atherosclerosis in the LDL receptor-deficient mouse in part through antiproliferative and anti-inflammatory actions. Am J Physiol Heart Circ Physiol 2007;293:H2394-402.
- Dupasquier CM, Weber AM, Ander BP, et al. Effects of dietary flaxseed on vascular contractile function and atherosclerosis during prolonged hypercholesterolemia in rabbits. Am J Physiol Heart Circ Physiol 2006;291:H2987-96.
- Thomson AB. Influence of dietary modifications on uptake of cholesterol, glucose, fatty acids, and alcohols into rabbit intestine. Am J Clin Nutr 1982;35:556-65.
- Thomson AB KM, Tavernini M. Early feeding of a high-cholesterol diet enhances intestinal permeability to lipids in rabbits. Pediatr Res 1987;21:347-51.
- Hanhoff T, Lucke C, Spener F. Insights into binding of fatty acids by fatty acid binding proteins. Mol Cell Biochem 2002;239:45-54.
- Burdge GC. Metabolism of alpha-linolenic acid in humans. Prostaglandins Leukot Essent Fatty Acids 2006;75:161-8.
- Bassett CM, McCullough RS, Edel AL, Patenaude A, LaVallee RK, Pierce GN. The alpha-linolenic acid content of flaxseed can prevent the atherogenic effects of dietary trans fat. Am J Physiol Heart Circ Physiol 2011;301:H2220-6.
- Prasad K. Dietary flax seed in prevention of hypercholesterolemic atherosclerosis. Atherosclerosis 1997;132:69-76.
- Francis AA, Austria JA, La Vallee RK, et al. The effects of dietary flaxseed on atherosclerotic plaque regression. Am J Physiol Heart Circ Physiol 2013;304:H1743-H1751.
- Talom RT, Judd SA, McIntosh DD, McNeill JR. High flaxseed (linseed) diet restores endothelial function in the mesenteric arterial bed of spontaneously hypertensive rats. Life Sci 1999;64:1415-25.
- Ander BP, Weber AR, Rampersad PP, Gilchrist JS, Pierce GN, Lukas A. Dietary flaxseed protects against ventricular fibrillation induced by ischemia-reperfusion in normal and hypercholesterolemic rabbits. J Nutr 2004;134:3250-6.
- Billman GE, Kang JX, Leaf A. Prevention of ischemia-induced cardiac sudden death by n-3 polyunsaturated fatty acids in dogs. Lipids 1997;32:1161-8.
- McLennan PL. Relative effects of dietary saturated, monounsaturated, and polyunsaturated fatty acids on cardiac arrhythmias in rats. Am J Clin Nutr 1993;57:207-12.
- Ander BP, Hurtado C, Raposo CS, et al. Differential sensitivities of the NCX1.1 and NCX1.3 isoforms of the Na+-Ca2+ exchanger to alpha-linolenic acid. Cardiovasc Res 2007;73:395-403.
- Guerin P, Bigot E, Patrice T. Evidence for antioxidants consumption in the coronary blood of patients with an acute myocardial infarction. J Thromb Thrombolysis 2013;35:41-7.
- Maddika S, Elimban V, Chapman D, Dhalla NS. Role of oxidative stress in ischemia-reperfusion-induced alterations in myofibrillar ATPase activities and gene expression in the heart. Can J Physiol Pharmacol 2009;87:120-9.
- Dhalla NS, Golfman L, Takeda S, Takeda N, Nagano M. Evidence for the role of oxidative stress in acute ischemic heart disease: A brief review. Can J Cardiol 1999;15:587-93.
- Prasad K. Hypocholesterolemic and antiatherosclerotic effect of flax lignan complex isolated from flaxseed. Atherosclerosis 2005;179:269-75.
- Prasad K. Hydroxyl radical-scavenging property of secoisolariciresinol diglucoside (SDG) isolated from flax-seed. Mol Cell Biochem 1997;168:117-23.
- Oomen CM, Feskens EJ, Rasanen L, et al. Fish consumption and coronary heart disease mortality in Finland, Italy, and The Netherlands. Am J Epidemiol 2000;151:999-1006.
- Ascherio A, Rimm EB, Giovannucci EL, Spiegelman D, Stampfer M, Willett WC. Dietary fat and risk of coronary heart disease in men: Cohort follow up study in the United States. BMJ 1996;313:84-90.
- Hu FB, Stampfer MJ, Manson JE, et al. Dietary intake of alphalinolenic acid and risk of fatal ischemic heart disease among women. Am J Clin Nutr 1999;69:890-7.
- Djoussé L, Pankow JS, Eckfeldt JH, et al. Relation between dietary linolenic acid and coronary artery disease in the National Heart, Lung, and Blood Institute Family Heart Study. Am J Clin Nutr 2001;74:612-9.
- Albert CM, Oh K, Whang W, et al. Dietary alpha-linolenic acid intake and risk of sudden cardiac death and coronary heart disease. Circulation 2005;112:3232-8.
- Burr ML, Fehily AM, Gilbert JF, et al. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and Reinfarction Trial (DART). Lancet 1989;2:757-61.
- de Lorgeril M, Renaud S, Mamelle N, et al. Mediterranean alphalinolenic acid-rich diet in secondary prevention of coronary heart disease. Lancet 1994;343:1454-9.
- Rodriguez-Leyva D, Weighell W, Edel AL, et al. Potent antihypertensive action of dietary flaxseed in hypertensive patients. Hypertension 2013;62:1081-9.
- Caligiuri SPB, Aukema HM, Ravandi A, Guzman R, Pierce GN. Flaxseed consumption reduces blood pressure in hypertensive patients by altering circulating oxylipins via an alpha linolenic acidinduced inhibition of soluble epoxide hydrolase. Hypertension 2014;64:53-9.
- *Corresponding Author:
- Dr Grant N Pierce
St Boniface Hospital Research Centre, 351 Tache Avenue, Winnipeg, Manitoba R2H 2A6.
Telephone: 204-235-3206
Fax: 204-235-0793
E-mail: gpierce@sbrc.ca
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact support@pulsus.com
Abstract
Cardiovascular disease remains a leading cause of mortality worldwide. It is now clear that myriad modifiable risk factors cause the majority of serious and chronic cardiovascular diseases. Most of these risk factors can be modified through changes in lifestyle and/or with pharmacotherapy. Advances in drug therapy for cardiovascular disease have had a large impact on heart disease over the past six decades. Currently, however, scientific investigations have expanded their focus to study the potential for foods and nutrients that provide the body with health benefits beyond normal nutrition. Many of these ‘functional foods’ or ‘nutraceuticals’ have demonstrated direct benefit for cardiovascular health – for both primary and secondary prevention of cardiovascular disease. Flaxseed is an example of a functional food that has demonstrated significant cardioprotective effects such as lowering the risk of sudden cardiac death or recurrent myocardial infarction. It has demonstrated antiarrhythmic, antihypertensive, antioxidant and lipid-lowering properties. Many of these beneficial effects have been attributed to the rich content of alpha-linolenic acid found in flaxseed. The effects of alpha-linolenic acid on cardiovascular disease, either delivered as a nutraceutical extract or through ingestion of flaxseed in the diet, are discussed in detail.
-Keywords
Alpha linolenic acid; Cardiovascular disease; Flaxseed; Heart disease; Polyunsaturated fatty acids
Functional Foods for the Prevention and Treatment of Cardiovascular Disease
Many natural food products possess a significant source of health benefit beyond normal nutrition. In recent years, a field of study has emerged that focuses on elucidating the beneficial or deleterious effects that specific foods and diets may have on various disease processes. One area of investigation is the role that specific nutrients play in the prevention of chronic cardiovascular disease (CVD) [1]. Largescale animal and human trials continue to identify the central importance of how different food sources affect modifiable cardiac risk factors such as diabetes, obesity and inflammation [2,3], diseases that are now reaching epidemic proportions.
One example of a diet-based clinical study that has changed foodintake guidelines to benefit cardiovascular health is the Dietary Approaches to Stop Hypertension (DASH) trial [4]. The DASH trial compared a typical Western diet with a diet rich in fruits, vegetables, low-fat dairy products and low in saturated fats. After two weeks of intervention, systolic blood pressure decreased significantly by 5.5 mmHg compared with the control Western diet [4,5]. This study demonstrates how simple alterations to a typical Western diet may result in a direct, positive net effect on health and the reduction of cardiovascular risk factors. Diet may also have significant implications in both the costs and benefits of long-term human health. As a result, many natural products that contain myriad properties that may directly influence chronic disease processes have been identified. Natural products that have been proven to elicit positive health effects are termed either ‘nutraceuticals’ or ‘functional foods’. As defined by Health Canada, a nutraceutical is “a product isolated or purified from foods that is generally sold in medicinal forms not usually associated with food. A nutraceutical is demonstrated to have a physiological benefit or provide protection against chronic disease”, whereas a “functional food is similar in appearance to, or may be, a conventional food, is consumed as part of a usual diet, and is demonstrated to have physiological benefits and/or reduce the risk of chronic disease beyond basic nutritional functions” [6]. Several nutraceuticals and functional foods have been shown to improve cardiovascular health [7,8].
The Role of Fibre and Omega-3 Fatty Acids
A product that has been clearly implicated in the reduction of cardiovascular risk is dietary fibre. Fibre possesses lipid-lowering, blood pressure- lowering and antioxidant effects [9,10]. Fibre is ingested, but is a nonabsorbable substance. Fibre may be ingested in either a soluble or insoluble form. Soluble fibre absorbs water, gaining viscosity and becomes fermented in the distal colon, while insoluble fibre binds gut contents, preventing absorption and increasing fecal mass [11,12]. Overall, dietary fibre interferes with the absorption of gut contents and increases gastric emptying. For example, fibre exhibits a cholesterollowering effect via its action as a bile salt sequestrant [13]. Soluble fibre binds bile salts in the gut, preventing their re-entry into circulation and eventual hepatic reabsorption; these salts are then excreted. The subsequent decrease in circulating bile salts causes an increase in expression of the hepatic enzyme alpha-cholesterol hydroxylase that transforms cholesterol to cholic acid to replenish lost bile salts [10]. This process further decreases the amount of endogenous cholesterol available for packaging and its entry into the circulation as a lipoprotein.
Recent findings suggest that the fibre viscosity may, in fact, be more closely linked with the lipid-lowering effects of dietary fibre. Vuksan et al [14] demonstrated that the lipid-lowering action of dietary fibre was significantly increased when a highly viscous form of fibre was administered in conjunction with a typical North American diet. This cholesterol-lowering effect was significantly greater than two other treatment arms containing greater amounts but less viscous forms of dietary fibre [14]. Contemporary guidelines suggest the ingestion of 30 g to 40 g of dietary fibre daily for a positive cardiovascular effect to be observed. The best dietary sources of fibre include whole fruits, vegetables, seeds and pulse crop products such as beans [15].
An example of a seed that provides a significant source of fibre is flaxseed (Linum usitatissimum). Flaxseed is composed of three major components: seed, lignin and oil. The fibre components found within the seed and lignin have been attributed to the modest cholesterollowering effects observed with the ingestion of flaxseed [16]. In total, dietary fibre accounts for 28% of flaxseed by weight [17]. Flaxseed has been identified as a functional food, and has been implicated in the reduction of primary cardiovascular risk and possible secondary prevention of CVD. Beyond flaxseed’s fibre component, it is believed to be beneficial for cardiovascular health due to its high concentration of the omega-3 polyunsaturated fatty acid (PUFA) alpha-linolenic acid (ALA). Flaxseed is one of the richest dietary sources of ALA [18]. Approximately 50% of the oil component of flax is ALA.
Omega-3 Fatty Acid Structure and Metabolism
In terms of chemical structure, omega-3 fatty acids are long-chain fatty acids chemically configured in a cis conformation and contain two double bonds within the chain. The final double bond is located at the third-last carbon of the chain. Omega-3 fatty acids are complex PUFAs that require more energy to make bioavailable than various other food molecules such as carbohydrates or saturated fats. As a result, omega-3 fatty acids have a much slower rate of metabolism. Omega-3 fatty acids are structurally similar to their omega-6 fatty acid counterparts. Omega-6 fatty acids, however, are easily converted to arachadonic acid. From arachadonic acid, a series of proinflammatory downstream products are synthesized such as prostaglandins and thromboxanes (Figure 1). Omega-3 and omega-6 fatty acids are not interconverted within humans because of the lack of the proper desaturase enzyme. As a result, omega-3 and omega-6 fatty acids compete for enzymatic metabolism with other PUFAs and eventually eicosinoids, which may confer either pro- or anti-inflammatory downstream effects (Figure 1). Under normal circumstances, omega-3 fatty acids are actually preferentially bound by metabolizing enzymes.
Figure 1: Biochemical pathway of ω-6 and ω-3 fatty acids. AA
Arachidonic acid; ALA α-linolenic acid; COX Cycloxygenases; DGLA
Dihomo γ-linolenic acid; DHA Docosahexaenoic acid; DPA
Docosapentaenoic acid; EPA Eicosapentaenoic acid; GLA γ-linolenic acid;
LA Linoleic acid; LOX Lipoxygenases; LTB4 Leukotriene B4; PGE2 Prostaglandin E2; PGI2 Prostaglandin I2; PGE3 Prostaglandin E3; PGI3 Prostaglandin I3; LTB5 Leukotriene B5; TXA2 Thromboxane A2
While ancient diets provided an omega-6 to omega-3 ratio of approximately one, today’s guidelines suggest a ratio of 4:1 for maintenance of good cardiovascular health. Many estimates, however, place current ratios of omega-6 to omega-3 approaching 20:1 in a typical Western diet [19]. This provides the body with an exponentially larger amount of omega-6 fatty acids than omega-3s. As a result, the abundance of omega-6 fatty acids overcomes the preferential binding of omega-3 fatty acids by metabolizing enzymes. An increased amount of omega-6 fatty acids become bound and form myriad proinflammatory eicosanoid molecules. Over time, this process may significantly increase an individual’s overall inflammatory state which, in turn, has the potential to significantly accelerate chronic disease processes such as atherosclerosis. Omega-3 fatty acids actually provide an opposing effect when metabolized. While ALA possesses independent antiinflammatory effects [20-22], it may also be metabolized to other PUFAs such as eicosapentaenoic acid (EPA) and, to a much lesser extent, docosahexaenoic acid (DHA) (Figure 1). These PUFAs stimulate the synthesis of eicosanoids, which decrease platelet adhesion and thrombosis [23,24].
Thus, in contrast to omega-6 fatty acids, omega-3 fatty acids confer anti-inflammatory characteristics and have been shown to clinically lower circulating markers of inflammation such as C-reactive protein and serum amyloid A [25,26]. The ill-proportioned ratio of omega-6 to omega-3 may also be responsible for the negative or null results obtained in recent clinical trials investigating possible effects on CVD. In fact, the omega 6:omega 3 ratio is a possible source of error in the results of the recent Alpha Omega trial [27]. In this dietary study, a margarine supplement rich in various omega-3 fatty acids was administered to postmyocardial infarction (MI) patients undergoing modern optimal medical therapy [27]. No positive beneficial cardiovascular effects were observed; however, the omega-6:omega-3 ratio in the margarine supplement was very high – in fact, it was 2.3 times higher than the ratio utilized in the Lyon Heart Diet study [27]. In light of this fact, added to the possibility that the Alpha Omega trial may have been underpowered, the results may become less significant. It is quite possible that the high omega-6 component of the dietary intervention may have masked the cardioprotective effects of the low-dose omega-3 fatty acid supplementation. Many of the large clinical trials that demonstrated a potential benefit of omega-3 fatty acids for secondary prevention of CVD also achieved a concomitant lowering of the omega-6:omega-3 ratio [28,29]. Thus, the ability of omega-3 fatty acids to play a significant role in the secondary prevention of CVD remains a viable possibility.
Omega-3 Fatty Acids and CVD
Omega-3 fatty acids may be obtained from several dietary sources. The most common source is fish and marine products. Fish provide an excellent source of the long-chain omega-3 fatty acids EPA and DHA. In 1973, Bang et al [30] first reported the beneficial effects of a marine based diet on cardiovascular health [30]. EPA and DHA obtained from marine sources have been demonstrated to reduce the risk of MI, sudden cardiac death and all-cause mortality [31,32]. The positive effects on cardiovascular health from omega-3 fatty acids derived from fish has prompted investigators to explore the potential benefits of other omega-3 fatty acids. Omega-3 fatty acids such as the short-chain fatty acids derived from plant sources may elicit similar protective effects and serve as an available alternative for people with allergies or general intolerance to fish. Increasing concerns about the contamination of marine products with elevated levels of substances toxic to the humans, such as mercury (particularly in commercially concentrated fish oils), has made the identification of alternative sources of omega-3 fatty acids even more important.
One example of a plant source rich in an omega-3 fatty acid is flaxseed. Flaxseed is also called linseed. Canada is one of the largest producers of flaxseed. Flaxseed is unique in that it is one of the richest sources of ALA. ALA is another omega-3 PUFA, apart from EPA and DHA, which has been independently identified as possessing several cardioprotective properties. Flaxseed exhibits cholesterol-lowering, antiarrhythmic, antiatherogenic and blood pressure-lowering effects [18,33].
It is believed that many of these beneficial effects may be due to its high content of ALA. Many of these effects have been well-demonstrated in various species of animals, as well as in healthy individuals. Current research is expanding the benefits of ALA to patients with pre-existing cardiovascular disease. This research will help to define the role of ALA beyond primary prevention of CVD and elucidate the possibility of ALA therapy for secondary treatment of CVD. The FlaxPAD study is a recent example of a local, large-scale clinical trial designed to determine the health effects of long-term dietary supplementation with flaxseed in patients with peripheral artery disease [34].
Absorption Characteristics and Bioavailability of ALA
Essential fatty acids, such as ALA, must be ingested and absorbed because the body does not have the capacity to synthesize them. Given the benefits of increasing circulating ALA levels in blood and tissues, it is important to identify mechanisms that may influence the absorption and overall bioavailability of ALA.
Presently, there is limited information about the characteristics of ALA absorption. Our laboratory, however, has elucidated the dosage and form of flaxseed that provides the greatest bioavailability of ALA in humans. In one study [35], three types of flax-supplemented muffins, containing either whole or milled flaxseed, or flax oil and each providing 6 g of ALA were administered daily to a population 18 to 49 years of age over a 12-week period. It was determined that flax oil provided the greatest bioavailability of ALA over 12 weeks of supplementation compared with whole or milled flaxseed. Another study from our laboratory demonstrated a significant rise in plasma ALA concentrations following only four weeks of flax oil supplementation [36]. Milled flaxseed, however, provided a similar bioavailability of ALA and was deemed to be significantly more tolerable over the longer 12-week supplementation period [35]. Furthermore, milled flaxseed does not degrade as quickly as flax oil and appears to be more easily integrated into an individuals’s daily diet. The bioavailability of ALA obtained from ground flaxseed is also unaffected by a subject’s age. Plasma ALA concentrations remained similar when comparing a young adult (18 to 29 years of age) and older adult (45 to 69 years of age) human population [37].
It remains largely unclear what effect interactions with other substances within the gut or enterocytes may have on the absorption and metabolism of ALA and other omega-3 fatty acids. Supplementation of the diet with cholesterol has led to an approximately 120-fold increase in plasma ALA levels in animals [38,39]. The increase in intestinal cholesterol is believed to stimulate ALA absorption through the intestinal wall [40,41]. However, essential fatty acids are not absorbed directly with cholesterol. While sterol transport occurs via the NPC1L1 transporter in the intestinal brush border in the gut, fatty acids are primarily absorbed passively via fatty acid binding transport proteins along the intestinal brush border in the small intestine [42]. Under normal conditions, essential fatty acids have a very high rate of absorption, exceeding 90% [43]. A potential mechanism for the observed increase in fatty acid absorption in the presence of increased gut cholesterol may be based on an increase in micelle formation and overall affinity and/or interaction of the intestinal brush border and gut contents [40,41]. Thus, conversely, molecules that interfere with the formation of micelles or the affinity of gut contents for the intestinal brush border may markedly affect the overall bioavailability of cardioprotective fatty acids.
Animal Research Demonstrating the Cardioprotective Effects of ALA
In recent years, several animal studies have demonstrated several cardioprotective effects of ALA [18,24]. These have included effects on the vasculature as well as directly on the heart. With respect to the former effects, Bassett et al [18,44] demonstrated in an LDL receptor −/− mouse model that the ingestion of flaxseed significantly attenuated the development of atherosclerosis induced by a diet either rich in cholesterol or industrial trans-fats. Prasad [45] also demonstrated the ability of flaxseed to reduce the amount of atherosclerotic plaque formations in hypercholesterolemic rabbits by 46% when ingested over eight weeks despite no change in serum lipid levels over that time. Recently, Francis et al [46] demonstrated that flaxseed not only slowed the progression of atherosclerosis, but also induced its regression once the plaques were established.
Flaxseed has also been associated with improved vascular function. The ability of an artery to contract or relax is mediated, in part, via systemic or paracrine signalling of the endothelial vessel lining. Dupasquier et al [39] determined that following eight weeks of dietary supplementation with cholesterol and flax in rabbits, the presence of 10% flax in the diet significantly reduced the progression of atherosclerosis compared with 0.5% cholesterol supplementation alone. Even the combined treatment of a diet containing both 0.5% cholesterol and 10% flax demonstrated the same antiatherogenic effect. This observation, however, was attenuated at 16 weeks. Despite this, flaxsupplemented groups still elicited a normalizing effect on aortic relaxation fully at eight weeks and partially at 16 weeks. These findings suggest a protective effect on endothelial-dependent vessel relaxation by flaxseed supplementation in hypercholesterolemic conditions [39]. Talom et al [47] also demonstrated in hypertensive rats that a diet rich in flaxseed improved acetylcholine-mediated endothelial relaxation. Disturbances in vascular function may be caused by an insult to the endothelium at the beginning of the atherosclerosis or the exacerbation of pre-existing atherosclerotic disease. The ability to positively affect vascular function may be one mechanism responsible for the beneficial effects of flaxseed in the primary prevention of CVD.
The direct effects of flaxseed on cardiac function have also been investigated. Ander et al [48] reported that the ingestion of flaxseed by rabbits over a 16-week period elicited an antiarrhythmic effect in hearts during an in vitro ischemia-reperfusion challenge. The presence of flax in the diet caused a decrease in ventricular fibrillation and a shortening of the QT interval either alone or in conjunction with cholesterol supplementation. Similar anti-arrhythmic effects have been reported for different species and with other omega-3 fatty acidbased interventions [49,50]. ALA may achieve its positive effects on postischemic cardiac function through an inhibition of Na+/Ca2+ exchange [51]. It may also achieve its effects through an antioxidative mechanism. Oxygen-derived free radicals are believed to participate in ischemic injury to the heart [52-54]. However, while the lignan component of flaxseed (secoisolariciresinol diglucoside [SDG]) has marked antioxidative effects [55,56], SDG is not absorbed from a flaxseedsupplemented diet. It is metabolized in the gut to enterolignans that can then circulate in the blood. Thus, the ultimate clinical relevance of SDG therapy remains unclear. This finding has not been translated into positive clinical trials [18].
Clinical Trials Demonstrating the Cardioprotective Effects of ALA
A number of large-scale clinical trials have supported the cardiovascular benefits of a diet rich in omega-3 fatty acids [17,29,33,57]. Several studies have implicated the significant role ALA supplementation may play in the primary prevention of CVD. Ascherio et al [58] studied 43,757 men 40 to 75 years of age and free of previous CVD or diabetes. After six years of follow-up, it was determined that following a multivariate analysis, ALA was inversely correlated with coronary artery disease. Furthermore, this effect was not demonstrated by marine omega-3 fatty acids such as EPA. Thus, ALA may function by an independent cardioprotective mechanism other than via its conversion to long-chain fatty acids such as EPA or DHA [58].
In a prospective, 10-year study involving 76,283 women, Hu et al [59] demonstrated that a long-term ingestion of a diet rich in ALA provided a significant protective effect against fatal MI in women who had no previous ischemic heart disease [59]. Djoussé et al [60] investigated the relationship between ALA intake and coronary artery disease in 4584 male and female study participants. ALA intake was significantly and inversely correlated with the prevalence OR (95% CIs) of coronary artery disease. Those subjects (male or female) with the highest ALA intake had the greatest reduction in risk [60].
Another large, population-based, prospective study conducted by Albert et al [61] analyzed the ability of ALA to protect against sudden cardiac death in 76,763 women. After 18 years of follow-up, the investigators determined that the quintiles of women with the two highest daily intakes of ALA had a 38% and 40% reduction in sudden cardiac death, respectively [61].
There is a growing body of evidence supporting the role of omega-3 fatty acids for the secondary prevention of CVD. For example, the Diet and Reinfarction Trial (DART) [62] was one of the first large clinical trials to investigate the potential role that omega-3 fatty acids play in the secondary prevention of CVD. The DART trial enrolled >2000 men who had recently experienced MI. Subjects were randomly assigned to one of three dietary interventions for two years. One treatment arm ingested fatty fish two to three times per week. After two years of dietary intervention and consultation, this group had a 29% reduction in all-cause mortality. Although there was no significant change in the primary end point of re-infarction or ischemic heart death among any of the groups, the marked increase in survival in the fatty fish group lead to the conclusion that a diet supplemented with fatty fish that are rich in omega-3 fatty acids may decrease risk of mortality in men following a MI [62].
The GISSI-Prevenzione study [29] was a large-scale prospective clinical trial with >11,000 study participants. Again, this trial enrolled patients who had recently experienced their first MI. The study demonstrated that over 3.5 years of intervention, a significant 10% decrease in relative risk of the primary end point of death, nonfatal MI or stroke was observed in the treatment group receiving regular omega-3 fatty acid supplementation. There was also a small, but significant reduction in circulating triglyceride levels in the omega-3 fatty acids group [29].
In terms of ALA, the Lyon Diet Heart Study [28] examined the cardioprotective role that a Mediterranean-based diet rich in omega-3 fatty acids, specifically ALA, may exhibit on the secondary prevention of CVD. Patients were randomly assigned to either control or dietary intervention during their hospital stay following their first MI. The study investigated the recurrence of secondary events such as cardiac death, recurrent MI, unstable angina and heart failure over a 46-month period. The Lyon Diet Heart Study demonstrated a protective effect of approximately 15% against a second event in patients receiving the dietary intervention for the duration of the study. Furthermore, ALA was the only fatty acid to be significantly associated with cumulative survival and the absence of a second MI at follow-up [28,63].
Given the multitude of cardioprotective properties elicited by omega-3 fatty acids, and ALA specifically, it is increasingly important to understand the factors that influence its mechanisms of action. Recent trials involving hypertensive patients supplemented with flaxseed may provide some answers. In a recent, placebo-controlled, double-blinded trial in which patients with peripheral arterial disease were administered foods that contained 30 g of milled flaxseed over one year, significant decreases in both systolic (15 mmHg) and diastolic (7 mmHg) blood pressure were observed in hypertensive patients [64]. This antihypertensive effect was associated with plasma ALA levels in these patients [64] and has been recently attributed to changes in the fatty acid-derived circulating oxylipin profile [65]. Further work to understand more fully the characteristics and mechanism responsible for the antihypertensive actions of dietary flaxseed are warranted and important in view of the powerful relationship between hypertension and significant clinical cardiovascular events.
Acknowledgements
This work is funded by the CIHR and St Boniface Hospital Research Foundation.
References
- Moore LL. Functional foods and cardiovascular disease risk: Building the evidence base. Curr Opin Endocrinol Diabetes Obes 2011;18:332-5.
- Alissa EM, Ferns GA. Functional foods and nutraceuticals in the primary prevention of cardiovascular diseases. J Nutr Metab 2012;2012:569486.
- Alberti KG, Eckel RH, Grundy SM, et al. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009;120:1640-5.
- Appel LJ, Moore TJ, Obarzanek E, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N Engl J Med 1997;336:1117-24.
- Champagne CM. Dietary interventions on blood pressure: The Dietary Approaches to Stop Hypertension (DASH) trials. Nutr Rev 2006;64:S53-6.
- Health Canada. Managing Health Claims for Foods in Canada: Towards a Modernized Framework, 1998. <http://www.hc-sc.gc.ca/ fn-an/label-etiquet/claims-reclam/nutra-funct_foods-nutra-fonct_ aliment-eng.php> (Accessed July 14, 2014).
- Mozaffarian D. Fish and n-3 fatty acids for the prevention of fatal coronary heart disease and sudden cardiac death. Am J Clin Nutr 2008;87:1991S-6S.
- Liu S, Manson JE, Lee IM, et al. Fruit and vegetable intake and risk of cardiovascular disease: The Women’s Health Study. Am J Clin Nutr 2000;72:922-8.
- Keys A, Grande F, Anderson JT. Fiber and pectin in the diet and serum cholesterol concentration in man. Proc Soc Exp Biol Med 1961;106:555-8.
- Sanchez-Muniz FJ. Dietary fibre and cardiovascular health. Nutr Hosp 2012;27:31-45.
- Anderson JW, Baird P, Davis RH Jr, et al. Health benefits of dietary fiber. Nutr Rev 2009;67:188-205.
- Roberfroid M. Dietary fiber, inulin, and oligofructose: A review comparing their physiological effects. Crit Rev Food Sci Nutr 1993;33:103-48.
- Dietary fiber as a binder of bile salts. Nutr Rev 1977;35:183-5.
- Vuksan V, Jenkins AL, Rogovik AL, Fairgrieve CD, Jovanovski E, Leiter LA. Viscosity rather than quantity of dietary fibre predicts cholesterol-lowering effect in healthy individuals. Br J Nutr 2011;106:1349-52.
- Mann J. Dietary carbohydrate: Relationship to cardiovascular disease and disorders of carbohydrate metabolism. Eur J Clin Nutr 2007;61(Suppl 1):S100-11.
- Prasad K. Flaxseed and cardiovascular health. J Cardiovasc Pharmacol 2009;54:369-77.
- Bloedon LT, Balikai S, Chittams J, et al. Flaxseed and cardiovascular risk factors: Results from a double blind, randomized, controlled clinical trial. J Am Coll Nutr 2008;27:65-74.
- Bassett CM, Rodriguez-Leyva D, Pierce GN. Experimental and clinical research findings on the cardiovascular benefits of consuming flaxseed. Appl Physiol Nutr Metab 2009;34:965-74.
- Simopoulos AP. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother 2002;56:365-79.
- Yoneyama S, Miura K, Sasaki S, et al. Dietary intake of fatty acids and serum C-reactive protein in Japanese. J Epidemiol 2007;17:86-92.
- Poudel-Tandukar K, Nanri A, Matsushita Y, et al. Dietary intakes of alpha-linolenic and linoleic acids are inversely associated with serum C-reactive protein levels among Japanese men. Nutr Res 2009;29:363-70.
- Erdinest N, Shmueli O, Grossman Y, Ovadia H, Solomon A. Anti-inflammatory effects of alpha linolenic acid on human corneal epithelial cells. Invest Ophthalmol Vis Sci 2012;53:4396-406.
- Kinsella JE, Lokesh B, Stone RA. Dietary n-3 polyunsaturated fatty acids and amelioration of cardiovascular disease: Possible mechanisms. Am J Clin Nutr 1990;52:1-28.
- Bloedon LT, Szapary PO. Flaxseed and cardiovascular risk. Nutr Rev 2004;62:18-27.
- Bemelmans WJ, Lefrandt JD, Feskens EJ, et al. Increased alphalinolenic acid intake lowers C-reactive protein, but has no effect on markers of atherosclerosis. Eur J Clin Nutr 2004;58:1083-9.
- Rallidis LS, Paschos G, Liakos GK, Velissaridou AH, Anastasiadis G, Zampelas A. Dietary alpha-linolenic acid decreases C-reactive protein, serum amyloid A and interleukin-6 in dyslipidaemic patients. Atherosclerosis 2003;167:237-42.
- Rauch B, Schiele R, Schneider S, et al. OMEGA, a randomized, placebo-controlled trial to test the effect of highly purified omega-3 fatty acids on top of modern guideline-adjusted therapy after myocardial infarction. Circulation 2010;122:2152-9.
- de Lorgeril M SP, Martin JL, Monjaud I, Delaye J, Mamelle N. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon Diet Heart Study. Circulation 1999;99:779-85.
- Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSIPrevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto miocardico. Lancet 1999;354:447-55.
- Bang HO, Dyerberg J, Hjoorne N. The composition of food consumed by Greenland Eskimos. Acta Med Scand 1976;200:69-73.
- Trikalinos TA, Lee J, Moorthy D, et al. Effects of eicosapentaenoic acid and docosahexanoic acid on mortality across diverse settings: Systematic review and meta-analysis of randomized trials and prospective cohorts: Nutritional Research Series, Vol 4. Rockville (MD), 2012.
- Marik PE, Varon J. Omega-3 dietary supplements and the risk of cardiovascular events: A systematic review. Clin Cardiol 2009;32:365-72.
- Rodriguez-Leyva D, Dupasquier CM, McCullough R, Pierce GN. The cardiovascular effects of flaxseed and its omega-3 fatty acid, alpha-linolenic acid. Can J Cardiol 2010;26:489-96.
- Leyva DR, Zahradka P, Ramjiawan B, Guzman R, Aliani M, Pierce GN. The effect of dietary flaxseed on improving symptoms of cardiovascular disease in patients with peripheral artery disease: Rationale and design of the FLAX-PAD randomized controlled trial. Contemp Clin Trials 2011;32:724-30.
- Austria JA, Richard MN, Chahine MN, et al. Bioavailability of alpha-linolenic acid in subjects after ingestion of three different forms of flaxseed. J Am Coll Nutr 2008;27:214-21.
- Kaul N, Kreml R, Austria JA, et al. A comparison of fish oil, flaxseed oil and hempseed oil supplementation on selected parameters of cardiovascular health in healthy volunteers. J Am Coll Nutr 2008;27:51-8.
- Patenaude A, Rodriguez-Leyva D, Edel AL, et al. Bioavailability of alpha-linolenic acid from flaxseed diets as a function of the age of the subject. Eur J Clin Nutr 2009;63:1123-9.
- Dupasquier CM, Dibrov E, Kneesh AL, et al. Dietary flaxseed inhibits atherosclerosis in the LDL receptor-deficient mouse in part through antiproliferative and anti-inflammatory actions. Am J Physiol Heart Circ Physiol 2007;293:H2394-402.
- Dupasquier CM, Weber AM, Ander BP, et al. Effects of dietary flaxseed on vascular contractile function and atherosclerosis during prolonged hypercholesterolemia in rabbits. Am J Physiol Heart Circ Physiol 2006;291:H2987-96.
- Thomson AB. Influence of dietary modifications on uptake of cholesterol, glucose, fatty acids, and alcohols into rabbit intestine. Am J Clin Nutr 1982;35:556-65.
- Thomson AB KM, Tavernini M. Early feeding of a high-cholesterol diet enhances intestinal permeability to lipids in rabbits. Pediatr Res 1987;21:347-51.
- Hanhoff T, Lucke C, Spener F. Insights into binding of fatty acids by fatty acid binding proteins. Mol Cell Biochem 2002;239:45-54.
- Burdge GC. Metabolism of alpha-linolenic acid in humans. Prostaglandins Leukot Essent Fatty Acids 2006;75:161-8.
- Bassett CM, McCullough RS, Edel AL, Patenaude A, LaVallee RK, Pierce GN. The alpha-linolenic acid content of flaxseed can prevent the atherogenic effects of dietary trans fat. Am J Physiol Heart Circ Physiol 2011;301:H2220-6.
- Prasad K. Dietary flax seed in prevention of hypercholesterolemic atherosclerosis. Atherosclerosis 1997;132:69-76.
- Francis AA, Austria JA, La Vallee RK, et al. The effects of dietary flaxseed on atherosclerotic plaque regression. Am J Physiol Heart Circ Physiol 2013;304:H1743-H1751.
- Talom RT, Judd SA, McIntosh DD, McNeill JR. High flaxseed (linseed) diet restores endothelial function in the mesenteric arterial bed of spontaneously hypertensive rats. Life Sci 1999;64:1415-25.
- Ander BP, Weber AR, Rampersad PP, Gilchrist JS, Pierce GN, Lukas A. Dietary flaxseed protects against ventricular fibrillation induced by ischemia-reperfusion in normal and hypercholesterolemic rabbits. J Nutr 2004;134:3250-6.
- Billman GE, Kang JX, Leaf A. Prevention of ischemia-induced cardiac sudden death by n-3 polyunsaturated fatty acids in dogs. Lipids 1997;32:1161-8.
- McLennan PL. Relative effects of dietary saturated, monounsaturated, and polyunsaturated fatty acids on cardiac arrhythmias in rats. Am J Clin Nutr 1993;57:207-12.
- Ander BP, Hurtado C, Raposo CS, et al. Differential sensitivities of the NCX1.1 and NCX1.3 isoforms of the Na+-Ca2+ exchanger to alpha-linolenic acid. Cardiovasc Res 2007;73:395-403.
- Guerin P, Bigot E, Patrice T. Evidence for antioxidants consumption in the coronary blood of patients with an acute myocardial infarction. J Thromb Thrombolysis 2013;35:41-7.
- Maddika S, Elimban V, Chapman D, Dhalla NS. Role of oxidative stress in ischemia-reperfusion-induced alterations in myofibrillar ATPase activities and gene expression in the heart. Can J Physiol Pharmacol 2009;87:120-9.
- Dhalla NS, Golfman L, Takeda S, Takeda N, Nagano M. Evidence for the role of oxidative stress in acute ischemic heart disease: A brief review. Can J Cardiol 1999;15:587-93.
- Prasad K. Hypocholesterolemic and antiatherosclerotic effect of flax lignan complex isolated from flaxseed. Atherosclerosis 2005;179:269-75.
- Prasad K. Hydroxyl radical-scavenging property of secoisolariciresinol diglucoside (SDG) isolated from flax-seed. Mol Cell Biochem 1997;168:117-23.
- Oomen CM, Feskens EJ, Rasanen L, et al. Fish consumption and coronary heart disease mortality in Finland, Italy, and The Netherlands. Am J Epidemiol 2000;151:999-1006.
- Ascherio A, Rimm EB, Giovannucci EL, Spiegelman D, Stampfer M, Willett WC. Dietary fat and risk of coronary heart disease in men: Cohort follow up study in the United States. BMJ 1996;313:84-90.
- Hu FB, Stampfer MJ, Manson JE, et al. Dietary intake of alphalinolenic acid and risk of fatal ischemic heart disease among women. Am J Clin Nutr 1999;69:890-7.
- Djoussé L, Pankow JS, Eckfeldt JH, et al. Relation between dietary linolenic acid and coronary artery disease in the National Heart, Lung, and Blood Institute Family Heart Study. Am J Clin Nutr 2001;74:612-9.
- Albert CM, Oh K, Whang W, et al. Dietary alpha-linolenic acid intake and risk of sudden cardiac death and coronary heart disease. Circulation 2005;112:3232-8.
- Burr ML, Fehily AM, Gilbert JF, et al. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and Reinfarction Trial (DART). Lancet 1989;2:757-61.
- de Lorgeril M, Renaud S, Mamelle N, et al. Mediterranean alphalinolenic acid-rich diet in secondary prevention of coronary heart disease. Lancet 1994;343:1454-9.
- Rodriguez-Leyva D, Weighell W, Edel AL, et al. Potent antihypertensive action of dietary flaxseed in hypertensive patients. Hypertension 2013;62:1081-9.
- Caligiuri SPB, Aukema HM, Ravandi A, Guzman R, Pierce GN. Flaxseed consumption reduces blood pressure in hypertensive patients by altering circulating oxylipins via an alpha linolenic acidinduced inhibition of soluble epoxide hydrolase. Hypertension 2014;64:53-9.