A Case of Two Bulbous Origins of Aortic Arch Branches and Compartmentalized Left Interscalene Triangle
Received: 08-Jul-2021 Accepted Date: Jul 28, 2021; Published: 04-Aug-2021, DOI: 10.37532/1308-4038.14(8).120-122
Citation: Tessema CB. A Case of Two Bulbous Origins of Aortic Arch Branches and Compartmentalized Left Interscalene Triangle. Int J Anat Var. 2021;14(8):120-122.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
During the dissection of an 83-year-old male cadaver, two bulbous origins of the aortic arch branches were noticed. The right divided into the brachiocephalic trunk and the left common carotid artery, and the left divided into the left vertebral and left subclavian arteries. A scalenus minimus muscle and a proximally split anterior scalene muscle divided the left interscalene triangle into four compartments through which the roots of the brachial plexus and the subclavian artery exited separately through each compartment. Variations like this could make the clinical distinction of aortic arch syndrome from thoracic outlet syndrome difficult, particularly, when the subclavian artery is involved and complicates the approach to structures in the interscalene triangle. Surgeons, radiologists, anesthesiologists and others involved in these areas should be cognizant of such variations in order to make the correct diagnosis, apply the appropriate procedure and institute the proper treatment for a better clinical outcome.
Keywords
Aortic arch; Bulbous origin; Scalenus minimus muscle; Interscalene triangle; Compartmentalization
Introduction
It is a common textbook knowledge that the aortic arch is a segment of the aorta that lies in the superior mediastinum above the transverse thoracic plane. It continues from the ascending aorta at the level of the right second sterncostal joint anteriorly and ends at the level of the left second sternocostal joint (intervertebral disc between T4 and T5) posteriorly to become descending thoracic aorta. Classically, the aortic arch gives three major branches (brachiocephalic trunk, left common carotid artery and left subclavian artery), which are collectively referred to as supra-aortic trunks, while the vertebral artery is frequently a branch from the left subclavian artery. The supra-aortic trunks supply the head and neck including the brain and spinal cord, upper limbs and their corresponding girdles, anterior and lateral thoracic wall and superior superficial back [1]. The structural and functional impairment of these branches in various diseases causes aortic arch syndrome. There are quite extensive and well documented variabilities in the branching patterns of these branches that occur during the embryonic development of pharyngeal arch arteries, which are significantly higher in women than in men [2]. This can be explained by the persistence of segments of the pharyngeal arch arteries that normally regress or by the regression of segments that should normally persist or both [3]. An angiographic study indicated that there is a higher incidence of common trunk for brachiocephalic trunk and left common carotid artery (bovine arch) [4]. A case report also showed right and left trunks arising from the aortic arch where the right is shared by the brachiocephalic trunk and left common carotid artery and the left is shared by the left vertebral and left subclavian arteries [5]. Another contrast enhanced computer tomographic study also noted three cases with common trunk for left vertebral and left subclavian arteries (vertebra-subclavian trunk), one of which had a common stem for brachiocephalic and left common carotid artery (bovine arch) [6]. Bovine arch is more common in black race and is associated with high incidence of diabetes mellitus and left sided stroke [7]. It is also believed that aortic arch variations are associated with various types of aortic diseases [8].
There are three well recognized primary scalene muscles (anterior, middle and posterior) with the triangular interscalene triangle (scalene triangle, interscalene gap) between the anterior scalene, the middle scalene and the first rib. As it was classically described, the roots of the brachial plexus and the subclavian artery exit through the interscalene triangles to distribute to the upper limbs and their corresponding girdles including the thoracic wall. Many conditions in this triangle like musculoskeletal variations, fibrous bands, tumors and posttraumatic conditions could result in vascular and/or neurogenic scalene anticus syndrome which is a component of thoracic outlet syndrome. There are a number of reports, describing the variable courses of these structures passing through the interscalene triangle like the brachial plexus passing between the middle and posterior scalene muscles rather than through the interscalene triangle, roots of the superior trunk of the brachial plexus passing through the anterior scalene muscles while the middle and inferior trunks of the brachial plexus and the subclavian artery pass through the interscalene triangle [9]. Developmentally, the scalene muscles arise from the pre-muscle tissue, which is likely to be from the paraspinal mesenchyme. By the 50th embryonic day, the three scalene muscles are fairly well differentiated, with their adult attachment to the cervical vertebrae and the ribs [10]. The persistence of certain muscle inclusions in the brachial plexus as well as groups of muscles that traverse elements of the plexus are related to the fragmentation of the original scalene pre-muscle mass as the limb buds develop [11]. Therefore, such errors occurring during the development of these muscles would result in variations of the muscles themselves and the structures passing between them. Another study pointed out that one of the rare muscular variations is the existence of the scalene minimums (scalene pleuralis) muscle that occurs with a wide range of incidences (7.8% - 71.1%) [12]. The presence of such muscle tissue in the interscalene triangle can be a cause of thoracic outlet syndrome [13].
Case Report
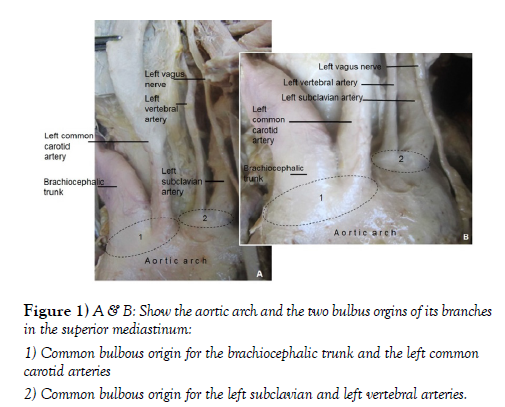
During careful dissection of an 83-years-old male cadaver, two major variations of practical importance were noted. The findings included a variant branching pattern of the aortic arch in the superior mediastinum and rare variations in the left root of the neck characterized by the presence of scalene minimums muscle and compartmentalized interscalene triangle. After careful further dissection and cleaning, pictures were taken for illustrations and the anonymous findings were given names for elucidation. The mediastinal dissection revealed two bulbus origins of the aortic arch branches. On the right side, the short common bulbous origin was shared by the brachiocephalic trunk and the left common carotid artery, while the relatively smaller of the two on the left side was shared by the left vertebral and left subclavian arteries (Figure 1).
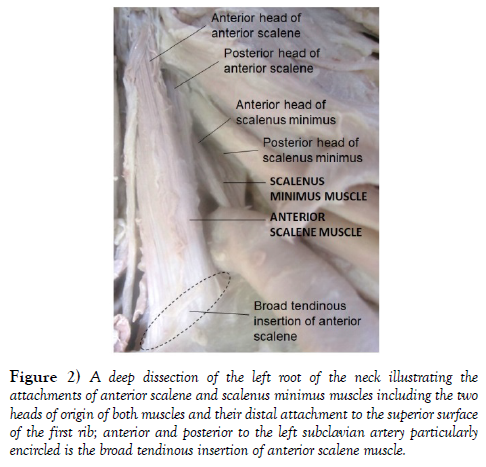
Dissection of the left root of the neck revealed a variant scalenus minimus muscle between the anterior and middle scalene muscles. The anterior scalene and scalenus minimus muscles were found to be having two heads of origin (anterior and posterior) from the C3 -C7 vertebrae. The anterior and posterior heads of anterior scalene muscle originated from the transverse processes of C3-C5 and C6 respectively and inserted by a broad flattened tendon to the superior surface of the first rib anterior to the subclavian sulcus.
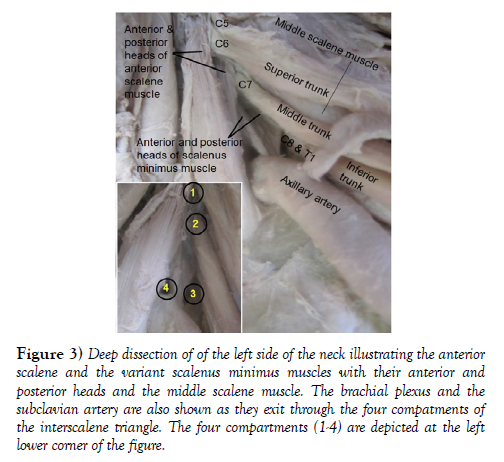
On the other hand, the anterior and posterior heads of the scalenus minimus muscle originated from the transverse processes of C6 and C7 respectively, where the anterior head ran deep to the posterior head of anterior scalene and merged with its posterior head before inserting to the superior surface of the first rib posterior to the subclavian sulcus (Figure 2). These two muscles with their corresponding two heads and the middle scalene muscle divided the interscalene triangle into four smaller triangular compartments. The first compartment, found between the anterior and posterior heads of the anterior scalene muscle, served the exit of the roots of the superior trunk of the brachial plexus (C5 & C6). The second compartment, between the anterior and posterior heads of the scalenus minimus muscle, served as an exit for the root of the middle trunk of the brachial plexus (C7). The third compartment, between the scalenus minimus, middle scalene and the first rib, served as an exit for the roots of the inferior trunk of the brachial plexus (C8 & T1). The fourth compartment, between the anterior scalene, scalenus minimus and the first rib, served the exit of the subclavian artery (Figure 3).
Figure 2: A deep dissection of the left root of the neck illustrating the attachments of anterior scalene and scalenus minimus muscles including the two heads of origin of both muscles and their distal attachment to the superior surface of the first rib; anterior and posterior to the left subclavian artery particularly encircled is the broad tendinous insertion of anterior scalene muscle.
Figure 3: Deep dissection of of the left side of the neck illustrating the anterior scalene and the variant scalenus minimus muscles with their anterior and posterior heads and the middle scalene muscle. The brachial plexus and the subclavian artery are also shown as they exit through the four compatments of the interscalene triangle. The four compartments (1-4) are depicted at the left lower corner of the figure.
Discussion
According to a computer tomographic study done on 2370 adult Chinese patients; 1985 (83.5%) had the classic branching pattern and 385 (16.2%) had variations of the aortic arch branches. Out of those with variations; 229 (9.6%) of them had a common trunk for the brachiocephalic trunk and the left common carotid artery, while 14 (0.6%) showed common origin of brachiocephalic and left common carotid artery combined with other additional variations [2]. An earlier review study also showed a high incidence (10-22%) of common origin of brachiocephalic trunk and left common carotid artery (bovine arch) [3]. In an angiographic study done on 633 patients; 527 (83%) cases of classic three branching pattern, 96 (15%) cases of two branches and many other rare variations were reported [4]. These three authors also noted left vertebral artery arising from the aortic arch between the left common carotid and left subclavian arteries but no common origin with left subclavian artery. However, an earlier case report documented a variation similar to the finding in this present case report, where two common trunks arose from the aortic arch, where the right trunk is shared by brachiocephalic trunk and left common carotid artery and the left by the left vertebral and left subclavian arteries [5]. Another contrast enhanced computer tomographic study on 410 patients similarly noted three cases of common trunks shared by the left vertebral and left subclavian arteries as vertebra-subclavian trunk (with an incidence of 0.73%), one of which also had a common stem for brachiocephalic and left common carotid artery (bovine arch) [6]. The present case differs from those reports in that there was no true or distinct trunk formation but the arteries shared two bulbous origins on the superior aspect of the aortic arch that could be comparable to the bovine aortic arch on the right side and vertebra-subclavian trunk on the left side. A retrospective study done on other group of 615 patients also showed that bovine aortic arch is more common in the black race (41%) and is associated with higher prevalence of diabetes mellitus and more leftsided strokes, concluding that there is a significant predilection of emboli (~30%) to the left cerebral hemisphere in patients with bovine arch. It was also pointed out that the aortic arch, though rare, is a site of various vascular pathologies like aneurysms, acute aortic syndromes, penetrating aortic ulcers, intramural hematomas, and possibly aortic dissection. These diseases may be more common in patients with aortic arch anomalies, particularly associated with bovine arches.
Various embryologic studies revealed that variabilities in the scalene muscles and the related structures occurs due to errors during development [9,10]. The variant scalenus minimus muscle is found to occur in wide range of incidences (7.8% - 71.1%) [12]. It was also emphasized that such variation is more important not only for anatomists, but also has clinical significance for the diagnosis of thoracic outlet syndrome and concluded that surgeons and anesthesiologists performing scalenectomy and interscalene brachial plexus block, respectively, should keep in mind the anatomical variations of this region [12]. The presence of such extra muscle further narrows the very much tight interscalene triangle predisposing structures in it to isolated or a more comprehensive neurovascular compression syndrome [13].
Though there are reports that closely resemble the vascular variations in this report, such combinations of bulbous origins of aortic arch branches, and the existence of scalenus minimus muscle associated with compartmentalized interscalene triangle by muscular slips were not found in any of the reviewed literature syndrome difficult, particularly, when the subclavian artery is involved. It should also be emphasized that the existence of the scalenus minimus and the compartmentalization of the interscalene triangle could be a cause of brachial plexopathy or isolated ventral nerve root and/or arterial compression syndromes in any of these narrow triangular spaces. This would also complicate preprocedural diagnosis, intraprocedural approach and postprocedural outcomes when dealing with the roots of the brachial plexus and the subclavian artery in the triangle. Therefore, surgeons, radiologists, anesthesiologists and others involved in carrying out various procedures in these areas should be cognizant of such variations in order to make the correct diagnosis, apply the suitable approach and institute the proper treatment for a better clinical outcome.
Acknowledgement
My thanks’ goes the donor and her family for their invaluable consent and body donation. I would also like to extend my thanks to the Department of Biomedical Science for its consistent encouragement and support. I am also grateful to Denelle Kees, Chelsy Swanson and John Opland for their assistance during the preparation and dissection of this cadaver.
REFERENCES
- Hegazy A. Clinical anatomy of thorax for medical students and doctors. LAP Lambert Academic Publishing, Egypt. 2018; 1:116.
- Wang L, Zhang J, Xin S. Morphologic features of aortic arch and its branches in adult Chinese population. J Vasc Surg. 2016; 64:1602-8.
- Kau T, Sinzig M, Gasser J, et al. Aortic development and anomalies. Semin Intervent Radiol. 2007; 24:141-152.
- Natsis KI, Tsitouridis IA, Didagelos MV, et al. Anatomical variation in the branching of the human aortic arch in 633 angiographies: Clinical significance and literature review. Surg Radiol Anat. 2009; 31: 319-23.
- Rameshbabu CS, Sharma V. Two common trunks arising from the arch of the aorta: Case report and literature review of a very rare variation. J Clin Diag Res. 2015;9: 05-07.
- Rameshbabu CS, Sharma V, Kumar A, et al. Vertebro-subclavian trunk: A rare aortic arch anomaly. J Clin Diag Res. 2018; 12:1-3.
- Matakas JD, Gold MM, Sterman J, et al. Bovine arch and stroke laterality. J Am Heart Assoc. 2020;9.
- Cheng SWK. Aortic arch pathologies- Incidence and natural history. Gefasschirurgie. 2016; 21:212-216.
- Haroun H. Cervical rib and thoracic outlet syndrome. MOJ Anat Physiol. 2016;2: 138-143.
- Radunovic M, Vukcevic B, Abramovic M, et al. Bilateral anatomic variation in the relation of the upper trunk of the brachial plexus to the anterior scalene muscle. Folia Morphol. 2019;78:195-198.
- Som PM, Laltman JT. Embryology, variations, and innervations of the human neck muscles. Neurographics. 2017; 7:215-242.
- Natsis KI, Totlis T, Didagelos M, et al. Scalenus minimus muscle: Overestimated or not? Anatomical study. Am Surg. 2013; 79:372-4.
- Lewis M, Prashar A, Toms AP, et al. The diagnosis of thoracic outlet syndrome. J Vasc Surg. 2014;2: 113-120.