Conservative management of extradural hematoma: A report of sixty-two cases
2 Department of Neurosurgery, West Virginia University, Morgantown, WV, USA, Email: bwold@mix.wvu.edu
Received: 13-Apr-2018 Accepted Date: Apr 23, 2018; Published: 30-Apr-2018
Citation: Zwayed ARH, Lucke-wold B. Conservative management of extradural hematoma: A report of sixty-two cases. 2018;2(2):05-09.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Background: Extradural hematomas (EDH) are considered life threatening in that the risk for brain herniation is significant. The current accepted understanding within the literature is to treat EDH via surgical evacuation of the hematoma.
Cases: In this case-series we report 62 cases of EDH managed conservatively without surgical intervention. Inclusion criteria were: Glasgow comma scale score 13-15, extradural hematoma confirmed by CT being less than 40 mm, less than 6 mm of midline shift, and no other surgical lesions present. Patients were initially observed in a surgical intensive care unit prior to discharge and had closely scheduled follow-up.
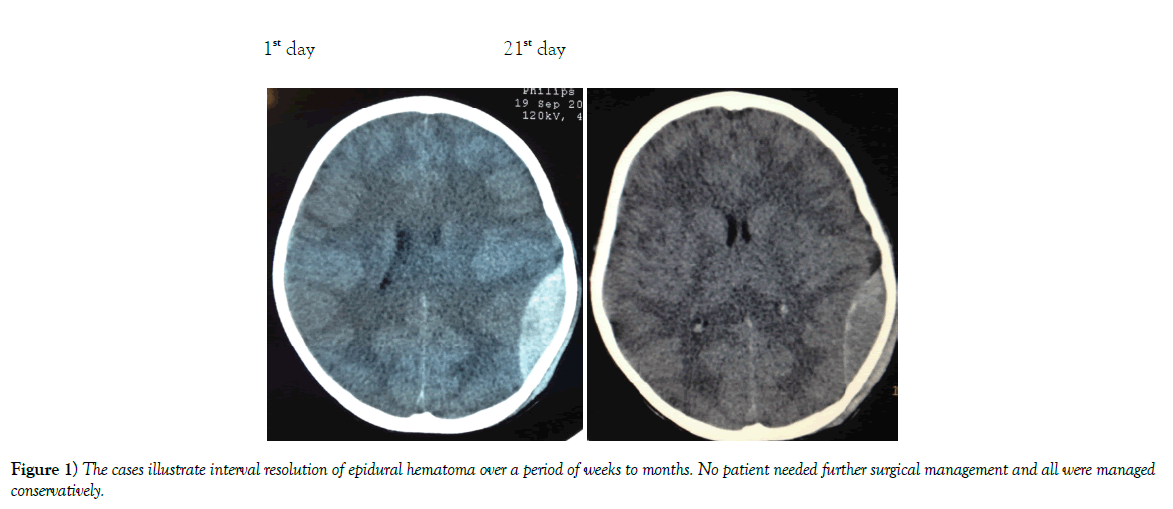
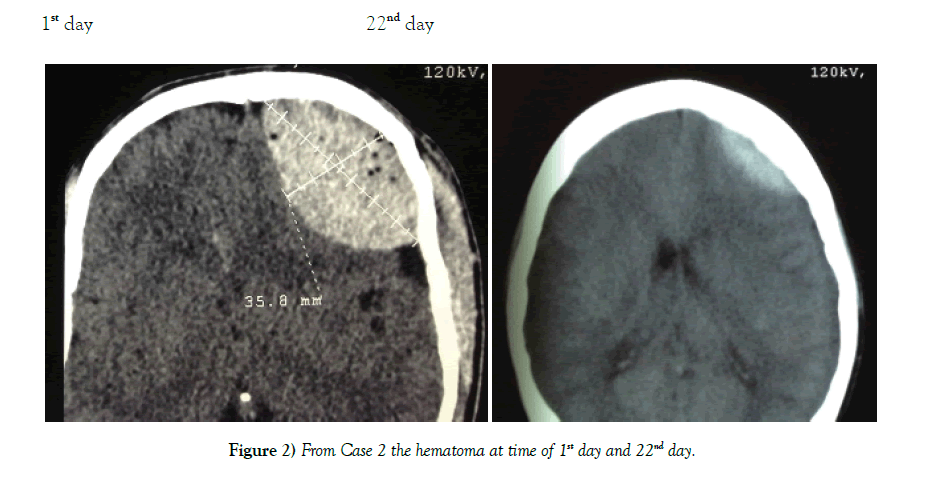
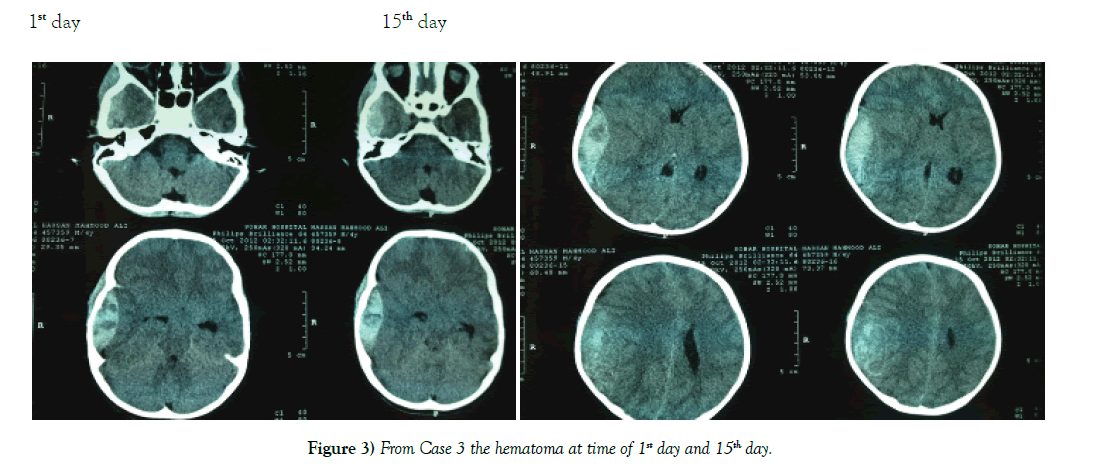
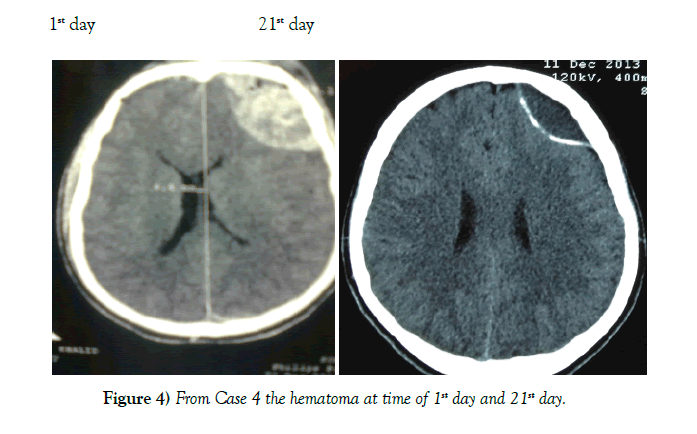
Results: Of the 62 cases none required emergent intervention and the majority had interval resolution of the epidural hematoma over time. Resolution was apparent by 21 days and definitive by 3 to 6 months.
Conclusion: Patients with EDH who have a high Glasgow comma scale score 13-15, volume <40 mm, and less than 6 mm of midline shift should be considered for conservative management. Our study indicates that these patients will have interval resolution of hematoma over time without worsening of symptoms.
Keywords
Epidural hematoma; Conservative management; Hemorrhagic volume; Glasgow coma scale; Herniation
Introduction
Extradural hematoma (EDH) accounts for 2% of all head injuries [1]. In selected patients conservative management may be a feasible option. Although several recent reports have described successful conservative management of epidural hematoma, surgical evacuation constitutes definitive treatment of this condition [2]. Evacuation of the hematoma, coagulation of bleeding sites, and inspection of the dura follows the craniotomy. The dura is then tented to the bone and occasionally; epidural drains are employed for 24-48 hours [3].
We report in this case series from Sohar Hospital 62 cases (from January 2013 to December 2017) that were managed conservatively without neurological intervention. Patients were males and females between 4-55 years old. Locations of the extradural hematomas were frontal, parietal, temporal, and occipital. Six cases were in the posterior fossa and one case was bilateral parietal. The thickness of these extra-dural hematomas was between 13-40 mm. These patients were discharged without neurological sequelae.
Conventionally, it has been taught that urgent evacuation is the accepted mode of management [4]. We argue based on our findings that with the routine use of Computer Tomography (CT) for management of head injury patients, non-operative management should be considered more often in select patients [5] (Figures 1-3).
Materials and Method
The study adhered to all Neurosurgical protocols and guidelines for head injury management in the Sultanate of Oman. Patients having a traumatic EDH with a Glasgow coma Score (GCS) of 13-15/15 were included in the study. Other inclusion criteria were: total EDH thickness less than 40 mm with midline shift or mass effect less than 6 mm, and no other surgical lesion on CT scan. Most of the cases were convexial EDH, but infra-tentoria EDH were also included (5 cases). All patients were monitored in a surgical high dependency care unit prior to discharge. Specifically GCS score, Pulse, Blood Pressure, and pupil diameter were observed.
Results
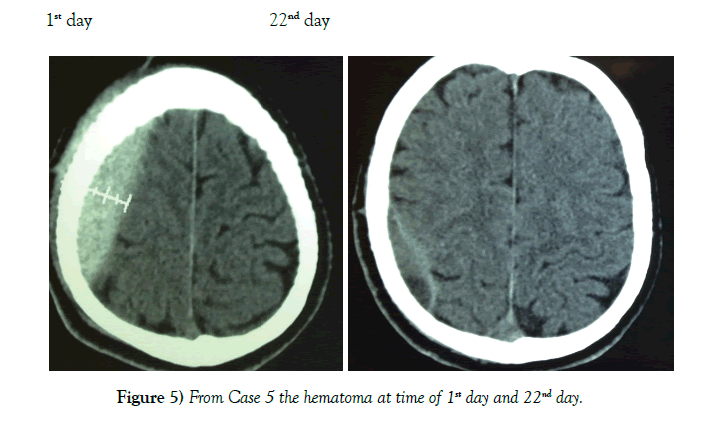
62 patients were successfully managed conservatively. Mode of injury was road traffic accidents for 49 cases, fall from a height for 10 cases, and assault in 3 cases. A summary of the cases is shown in Table 1. Patient population consisted of 51 males and 11 females between the ages of 4-55 years (Table 2). Location of the EDH was in the frontal region in 24 cases, parietal region in 17 cases, temporal region 12 cases, bi-parietal 1 case, parieto–occipital 3 cases, and posterior fossa area 5 cases (Table 3). The thickness of EDH ranged from 13 mm–40 mm. At no point of time did any of the 62 cases show any clinical signs of raised intracranial pressure. Immediate neurosurgical care was available at all times (Figures 4 and 5).
| No. | Age /yrs. | Sex | Size /mm | Mid line shift /mm | location |
|---|---|---|---|---|---|
| 1 | 4 | M | 15 | 2 | Left temporal |
| 2 | 10 | M | 26 | 3 | Left parietal |
| 3 | 12 | M | 28 | 4 | Right frontal |
| 4 | 16 | M | 15 | 2 | Left frontal |
| 5 | 17 | M | 17 | 4 | Left posterior fossa |
| 6 | 22 | M | 17 | 2 | Left temporal |
| 7 | 53 | M | 40 | 5 | Left frontal |
| 8 | 16 | M | 28 | 5 | Left parietal |
| 9 | 33 | M | 20 | 4 | Right temporal |
| 10 | 26 | M | 32 | 6 | Left frontal |
| 11 | 35 | M | 39 | 4 | Right frontal |
| 12 | 19 | M | 17 | 3 | Left temporal |
| 13 | 6 | M | 19 | 4 | Right posterior fossa |
| 14 | 25 | M | 34 | 5 | Left frontal |
| 15 | 21 | M | 35 | 4 | Left parietal |
| 16 | 37 | M | 33 | 3 | Right frontal |
| 17 | 24 | M | 28 | 4 | Right frontal |
| 18 | 31 | M | 29 | 0 | Bi-parietal |
| 19 | 44 | M | 28 | 5 | Left frontal |
| 20 | 18 | M | 32 | 4 | Left parietal |
| 21 | 32 | M | 27 | 6 | Right frontal |
| 22 | 27 | M | 22 | 3 | Right parietal |
| 23 | 17 | M | 14 | 3 | Left temporal |
| 24 | 15 | M | 37 | 5 | Left frontal |
| 25 | 28 | M | 19 | 3 | Right temporal |
| 26 | 21 | M | 16 | 3 | Right posterior fossa |
| 27 | 29 | M | 21 | 5 | Left parietal |
| 28 | 24 | M | 22 | 3 | Right parieto-occipital |
| 29 | 44 | M | 13 | 4 | Left temporal |
| 30 | 19 | M | 26 | 5 | Right parietal |
| 31 | 35 | M | 40 | 6 | Left frontal |
| 32 | 27 | M | 36 | 3 | Right frontal |
| 33 | 37 | M | 20 | 2 | Left parietal |
| 34 | 20 | F | 28 | 3 | Right parietal |
| 35 | 7 | F | 20 | 5 | Left frontal |
| 36 | 21 | F | 15 | 3 | Left temporal |
| 37 | 23 | F | 37 | 4 | Right frontal |
| 38 | 27 | F | 31 | 3 | Left frontal |
| 39 | 36 | F | 29 | 3 | Left parietal |
| 40 | 5 | M | 28 | 3 | Left frontal |
| 41 | 11 | M | 34 | 5 | Right temporal |
| 42 | 19 | M | 37 | 4 | Right posterior fossa |
| 43 | 15 | M | 27 | 5 | Left parietal |
| 44 | 23 | F | 37 | 6 | Left parieto-occipital |
| 45 | 19 | M | 32 | 5 | Left temporal |
| 46 | 34 | F | 33 | 4 | Right parietal |
| 47 | 27 | M | 38 | 6 | Left frontal |
| 48 | 36 | M | 23 | 2 | Right frontal |
| 49 | 9 | M | 40 | 5 | Left parietal |
| 50 | 24 | M | 35 | 5 | Right parietal |
| 51 | 22 | F | 30 | 4 | Left frontal |
| 52 | 35 | M | 27 | 3 | Left temporal |
| 53 | 29 | M | 26 | 3 | Left frontal |
| 54 | 18 | M | 27 | 5 | Right temporal |
| 55 | 33 | M | 25 | 4 | Right posterior fossa |
| 56 | 45 | M | 19 | 3 | Left parietal |
| 57 | 27 | M | 21 | 5 | Right parito-occipital |
| 58 | 11 | F | 17 | 3 | Left temporal |
| 59 | 19 | M | 31 | 5 | Right parietal |
| 60 | 23 | M | 20 | 3 | Left frontal |
| 61 | 50 | M | 22 | 3 | Right frontal |
| 62 | 28 | M | 32 | 5 | Left parietal |
Table 1: 62 patients were successfully managed conservatively. Mode of injury was road traffic accidents for 49 cases, fall from a height for 10 cases, and assault in 3 cases
| Age | Males | Females | Total |
|---|---|---|---|
| <10 years | 6 | 1 | 7 |
| 10-20 years | 17 | 1 | 18 |
| 21-30 years | 14 | 7 | 21 |
| 31-40 years | 9 | 2 | 11 |
| 41-50 years | 4 | 0 | 4 |
| >50 years | 1 | 0 | 1 |
| Total | 51 | 11 | 62 |
Table 2: Patient population consisted of 51 males and 11 females between the ages of 4-55 years
| Site | Right | Left | Total |
|---|---|---|---|
| Frontal | 9 | 15 | 24 |
| Temporal | 4 | 8 | 12 |
| Parietal | 6 | 11 | 17 |
| Posterior fossa | 4 | 1 | 5 |
| Bi-parietal | 1 | 1 | |
| Parieto occipital | 2 | 1 | 3 |
| 62 | |||
Table 3: Location of the EDH was in the frontal region in 24 cases, parietal region in 17 cases, temporal region 12 cases, bi-parietal 1 case, parieto–occipital 3 cases, and posterior fossa area 5 cases
Discussion
Patients with EDH who are conscious have a very low mortality [6]. Only patients with loss of consciousness are at risk for the serious complication of herniation [7]. Non-operative management of EDH has been well documented [8]. Patient selection is therefore of utmost importance in conservative management of EDH [9]. Various factors have been found to influence the management strategy. Dubey & Bezircioglu have recommended a thickness of EDH less than 30 mm for conservative management, Bullock found 12-38 mm suitable, whereas Giordano and colleagues have managed patients with a thickness up to 55 mm, without surgery [10]. We chose 40 mm for our cutoff.
Location is also an important consideration based on recovery rates. Most studies have taken only supratentorial hematomas into consideration [11]. Reports in the literature show a posterior fossa EDH volume less than 10 mm to be favorably managed conservatively [12]. Our study expands this criteria in that our patients recovered well from posterior fossa even up to 30 mm. Furthermore temporal EDH was unlikely to be managed conservatively historically as compared to frontal or parietal but here we have 12 cases that were managed conservatively. We therefore urge increased expansion for EDH locations that can be managed conservatively.
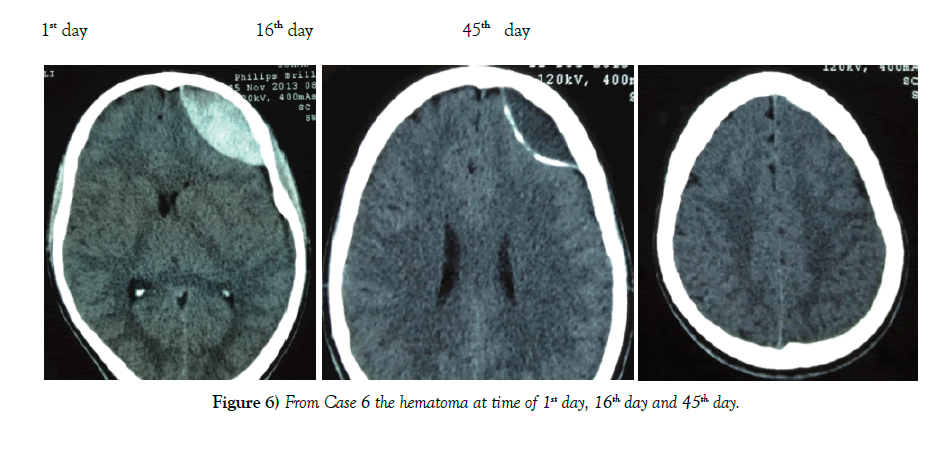
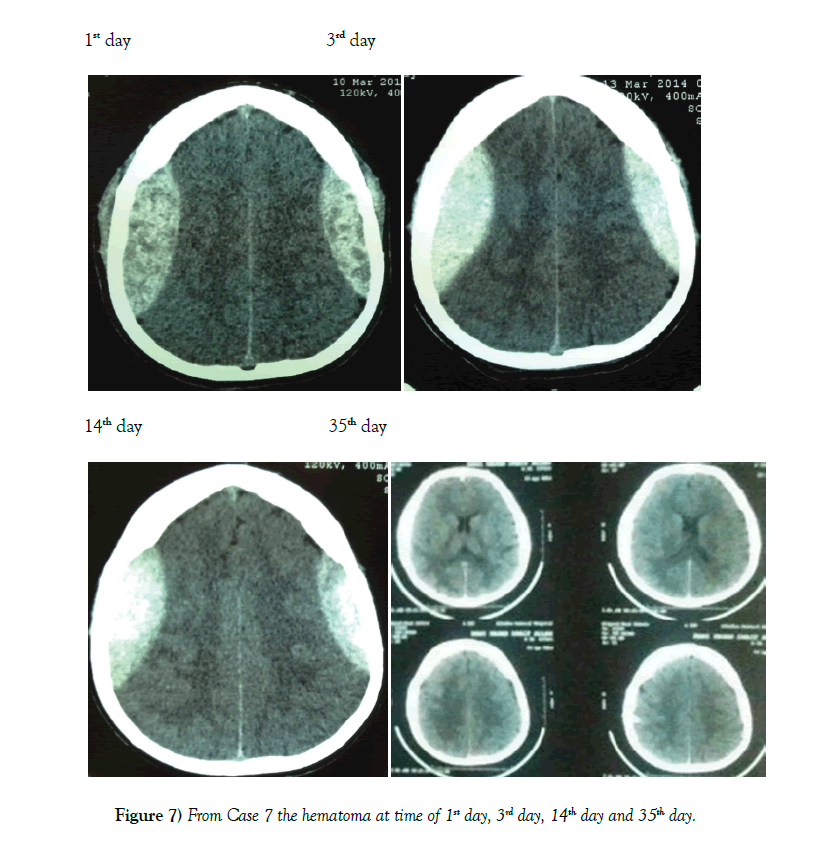
A lower GCS has been associated with a worse outcome in most studies [13]. We therefore chose the cutoff of GCS 13 or higher for our patients. Other factors like a thickness >15 mm and a midline shift >5 mm have also been found to unfavorably influence the outcome [14]. We chose the cutoff of 6 mm for midline shift. Another important consideration is the timing of when to get a repeat CT scan. Literature has shown that EDH enlargement occurs within 36 hours and a repeat CT is useful at this time [15]. EDH enlargement occurrs in 23% of patient and mean time to enlargement is within 8 hours of injury [16]. Repeat imaging is necessary to determine if patients are still eligible for conservative management (Figures 6 and 7).
If a lesion is small and the patient is in good neurological condition, observing the patient with frequent neurological examinations is reasonable [17]. Acute anterior temporal tip EDHs are one subset, which are likely to have a benign course and can usually be followed with imaging and observation [18]. The venous origins of these EDHs compared to the middle meningeal EDHs are a likely reason why they have slow expansion and eventual coagulation of the bleeding source making them more amenable to conservative therapy [19].
Conclusion
Although conservative management is often left to clinical judgment, the “Guidelines for the Surgical Management of Traumatic Brain Injury” recommended that patients who exhibit an EDH that is less than 15- mm thick and less than 5-mm midline shift, without a focal neurological deficit and GCS greater than 8 can be treated none operatively [20]. We have expanded this criteria to 40 mm thickness, GCS 13 or greater, and less than 6 mm midline shift. Early follow-up scanning should be used to assess a further increase in hematoma size prior to deterioration. This case series provides important guidance for neurosurgical care of patients with epidural hematomas.
REFERENCES
- Arrese I, Lobato RD, Gomez PA. Hyperacute epidural haematoma isodense with the brain on computed tomography. Acta Neurochir. 2004;146(2):193-4.
- Basamh M, Robert A, Lamoureux J, et al. Epidural Hematoma Treated Conservatively: When to Expect the Worst. Can J Neurol Sci. 2016;43(1):74-81.
- Bejjani GK, Donahue DJ, Rusin J, et al. Radiological and clinical criteria for the management of epidural hematomas in children. Pediatr Neurosurg. 1996;25(6):302-8.
- Bezircioglu H, Ersahin Y, Demircivi F, et al. Nonoperative treatment of acute extradural hematomas: analysis of 80 cases. J Trauma. 1996;41(4):696-8.
- Bozbuga M, Izgi N, Polat G, et al. Posterior fossa epidural hematomas: observations on a series of 73 cases. Neurosurg Rev. 1999;22(1):34-40.
- De Souza M, Moncure M, Lansford T, et al. Nonoperative management of epidural hematomas and subdural hematomas: is it safe in lesions measuring one centimeter or less?. J Trauma. 2007;63(2):370-2.
- Guay J. Estimating the incidence of epidural hematoma - is there enough information? Can J Anaesth. 2004;51(5):514-5.
- Guo C, Liu L, Wang B, et al. Swirl sign in traumatic acute epidural hematoma: prognostic value and surgical management. Neurol Sci. 2017;38(12):2111-6.
- Jamous MA, Aziz HA, Kaisy FA, et al. Conservative management of acute epidural haematoma in paediatric age group. Pediatr Neurosurgery. 2009;45:181-4.
- https://emedicine.medscape.com/article/1137065-treatment
- Hamilton M, Wallace C. Non operative management of acute epidural haematoma diagnosed by CT: the neuroradiologist`s role. Am J Neuroradiol. 1992;13:853-9.
- Huisman TA, Tschirch FT. Epidural hematoma in children: Do cranial sutures act as a barrier? J Neuroradiol. 2009;36(2):93-7.
- Irie F, Brocque RL, Kenardy J, et al. Epidemiology of traumatic epidural hematoma in younge age. J Trauma. 2011;71(4):847-53.
- Miller DJ, Steinmetz M, McCutcheon IE. Vertex epidural hematoma: surgical versus conservative management: two case reports and review of the literature. Neurosurgery. 1999;45(3):621-5.
- Narayan RK, Wilberger JE Jr, Povlishock JT, Eds. Neurotrauma. First ed. New York: McGraw Hill; 1996.
- Rivas JJ, Lobato RD, Sarabia R, et al. Extradural hematoma: analysis of factors influencing the courses of 161 patients. Neurosurgery. 1988;23(1):44-51.
- Sencer A, Aras Y, Akcakaya MO, et al. Posterior fossa epidural hematomas in children: clinical experience with 40 cases. J Neurosurg Pediatr. 2012;9(2):139-43.
- Servadei F. Prognostic factors in severely head injured adult patients with epidural haematoma's. Acta Neurochir. 1997;139(4):273-8.
- Toner A, Prabhu P. Reliable detection of epidural haematomas. Br J Anaesth. 2009;102(1):140-1.
- Yilmazlar S, Kocaeli H, Dogan S. Traumatic epidural haematomas of nonarterial origin: analysis of 30 consecutive cases. Acta Neurochir. 2005;147(12):1241-8.