Effect of atorvastatin on circulating adipocytokine levels in diabetic patients
- *Corresponding Author:
- Dr Alberto Francisco Rubio-Guerra
Motozintla # 30, Col Letran valle, México, DF CP 03600
Tel: 52-555-539-3584
Fax: 52-555-539-3584
E-mail: clinhta@hotmail.com
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http:// creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
[ft_below_content] =>Keywords
Adiponectin; Atorvastatin; Diabetic patients; Resistin
Adipose tissue has been previously described as an active endocrine organ, and adiponectin and resistin have been described as peptides that are secreted by adipocytes (adipocytokines). Both hormones modulate the effects of insulin. In addition, hypoadiponectinemia and hyper-resistinemia are associated with hypertension, obesity and type 2 diabetes [1]. It has also been reported that hypoadiponectinemia is a risk factor in the development of cardiovascular disease [2] and hyperresistinemia is associated with inflammation and the pathways that lead to atherosclerosis [3]. Rubio et al [1] demonstrated that the adiponectin/resistin index correlates with carotid intima-media thickness.
Statins are effective cholesterol-lowering drugs that reduce the risk for stroke and cardiovascular disease, especially in diabetic patients [4]. However, beyond their cholesterol-lowering effects, statins have lipid-independent (or pleiotropic) properties, including reduction of oxidative stress, normalization of endothelial function and antiinflammatory actions [5].
Unfortunately, the use of statins includes the potential risk for adverse effects, such as myopathy, rhabdomyolysis and increasing levels of transaminases. In addition, recent evidence suggests an increased risk for new-onset diabetes mellitus [6].
Several pharmacological agents affect the production of adipocytokines. Statins modulate the production of adipocytokines and may modify insulin sensitivity as a consequence of an increase or reduction in circulating adipocytokines [7]. In fact, pravastatin and pitavastatin increase serum adiponectin concentrations, whereas simvastatin decreases its circulating levels.
Contradictory results have been reported regarding the effect of atorvastatin on adiponectin levels. Some authors have found that atorvastatin increases the levels of this adipocytokine [8], whereas other studies have reported no change in total adiponectin in response to atorvastatin [9]. Most studies have found that statin therapy has little to no effect on circulating resistin levels [7].
The aim of the present study was to evaluate the effect of atorvastatin on adipocytokine levels in normotensive patients with type 2 diabetes in Mexico.
Methods
A total of 20 normotensive patients with type 2 diabetes mellitus of >12 months’ duration and without previous treatment with thiazolindinediones, statins or inhibitors of the renin-angiotensin system were included. All patients received 10 mg atorvastatin daily, for the duration of the three-month study.
Diagnosis of type 2 diabetes was made according to the American Diabetes Association criteria [10].
Venous samples were collected in the morning after a 12 h overnight fast at the beginning and the conclusion of the study for all patients. Duplicate samples of circulating levels of adiponectin and resistin were measured using ELISA (R&D Systems, USA) according to the manufacturer’s recommendations. ELISAs were performed by personnel blinded to the study. Intra-assay coefficient of variability (CV) for adiponectin was 3.4% and interassay CV was 5.8%. Whereas intra-assay CV for resistin was 5.5% and inter-assay CV was 9.2%. Fasting glycemia and glycated hemoglobin (HbA1c) levels were measured, and lipid profiles were recorded.
Patients with any of the following diagnoses were excluded from the study: heart, hepatic or renal failure; evidence of valvular heart disease; heart block or cardiac arrhythmia; acute coronary syndrome or cerebrovascular disease diagnosed six months before the study’s initiation; autoimmune disease; pregnancy; urinary tract infection; fever; or a history of alcohol abuse and/or psychotropic drugs.
The study was conducted with the approval of the research and medical ethics committee of the authors’ hospital, in accordance with the Declaration of Helsinki. Participants provided written informed consent before their inclusion in the study protocol.
Statistical analysis
Data are presented as the mean ± SD. Statistical analysis was performed using the Mann-Whitney U test; P<0.05 was considered to be statistically significant.
Results
Basline characteristics of the patients are presented in Table 1. For all patients, mean (± SD) low-density lipoprotein (LDL) levels were reduced (4.013±0.46 mmol/L to 2.72±0.32 mmol/L; P<0.01).
| Age, years | 54±9.2 |
| Sex, male/female, n/n | 8/12 |
| Glycemia, mmol/L | 7.54±1.48 |
| Glycated hemoglobin, % | 6 |
| Low-density lipoprotein, mmol/L | 4.013±0.46 |
| Body mass index, kg/m2 | 32.4±5.1 |
| Blood pressure, systolic/diastolic, mmHg | 122/72 |
| History of diabetes mellitus, years | 8.8 |
Data presented as mean or mean ± SD unless otherwise indicated
Table 1: Baseline patient characteristics
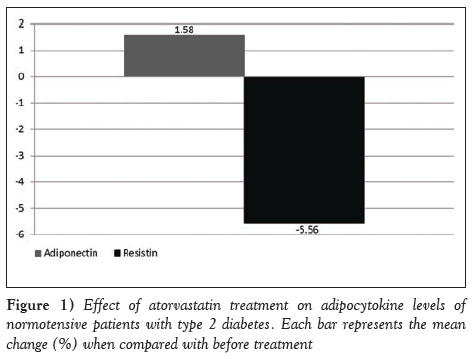
Atorvastatin modestly increased the levels of adiponectin by 1.67 μg/mL (6.85 μg/mL to 8.43 μg/mL [95% CI 1.4 to 4.7]; P=0.0602), and significantly decreased the levels of resistin by 5.51 pg/mL (15.65 pg/mL to 10.09 pg/mL [95% CI 3.92 to 6.83]; P=0.022) (Figure 1). None of the patients experienced adverse events.
Discussion
The Mexican National Survey on Health and Nutrition 2012 reported that for Mexicans >20 years of age, 32.4% were affected by obesity and 38.8% were overweight, contributing to a high prevalence of type 2 diabetes and cardiovascular disease in Mexico [11]. Due to the role of hypoadiponectinemia and hyperresistinemia in the development of insulin resistance and in the pathways that lead to atherosclerosis, both adipocytokines are potential treatment targets. In fact, increasing adiponectin and reducing resistin levels contribute to reducing the risk for cardiovascular disease [1,12].
In the present study, we found that therapy with 10 mg atorvastatin daily for 12 weeks significantly reduced LDL and resistin levels in normotensive patients with type 2 diabetes, whereas statin induced a modest, but insignificant, increase in adiponectin. It is important to note that in our study, circulating levels of adipocytokines were measured in duplicate; therefore, intraindividual variation was taken into account when statistical analyses were performed.
It is also important to note that our patients were naive to thiazolindinedione, statin and angiotensin-converting enzymes/angiotensin II receptor blockers, because these drugs have been shown to modify circulating levels of adipocytokines [1].
Statins have been demonstrated to reduce cardiovascular risk in diabetic patients due to their lipid dependent [4] and lipid independent (or pleiotropic) actions [13]. In addition, their use in type 2 diabetic patients is effective and safe; in fact, the American Diabetes Association recommends that statins should be added, regardless of lipid value, to the treatment regimens of diabetic patients with overt cardiovascular disease, or in individuals without cardiovascular disease but who are >40 years of age and have ≥1 cardiovascular risk factor(s) [10].
Our results are consistent with those reported by Von Eynatten et al [9], who found no change in total adiponectin in response to the statin in diabetic patients [9]; however, Ando et al [8] found a significant increase in the adipocytokine levels in 36 hypercholesterolemic patients. It is important to note that in the study by Von Eynatten et al [9], and in our study, all patients were diabetic, whereas in the study by Ando et al [8], only four (11%) patients were diabetic. Studies comparing the effect of atorvastatin on circulating adiponectin levels in diabetic and nondiabetic patients are needed.
To our knowledge, the present study is the first to report a significant reduction in circulating resistin levels with the use of 10 mg of atorvastatin, whereas others reports have reported no change in resistin levels with that dose [7]. We do not have an explanation for this difference, although ethnicity may play a role. In fact, Von Eynatten et al [14] have shown that 40 mg of atorvastatin significantly reduces the circulating levels of resistin.
The increased incidence of diabetes and obesity in Mexico is accompanied by a greater incidence of cardiovascular complications, and the use of lipid-lowering drugs is accompanied not only by reductions in LDL levels, but also by a reduction in cardiovascular risk due to their pleiotropic effects. Our results demonstrate, in part, the pathways that lead to some of those pleiotropic actions.
Conclusion
Our results demonstrate that atorvastatin significantly reduced the levels of resistin and induced a modest, yet insignificant, increase in adiponectin levels. This may explain, in part, the lipid independent cardiovascular effects observed with statins in diabetic patients.
References
- Rubio-Guerra AF, Cabrera-Miranda JL, Vargas-Robles H, et al. Correlation between levels of circulating adipokines and adiponectin/resistin index with carotid intima-media thickness in hypertensive type 2 diabetic patients. Cardiology 2013;125:150-3.
- Behre CJ. Adiponectin, obesity and atherosclerosis. Scand J Clin Lab Invest 2007;67:449-58.
- Lozano-Nuevo JJ, Estrada-Garcia T, Vargas-Robles H, et al. Correlation between circulating adhesion molecules and resistin levels in hypertensive type-2 diabetic patients. Inflamm Allergy Drug Targets 2011;10:27-31.
- Colhoun HM, Betteridge DJ, Durrington PN, et al. CARDS investigators. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): Multicentre randomised placebo-controlled trial. Lancet 2004;364:685-96.
- Profumo E, Buttari B, Saso L, Rigano R. Pleiotropic effects of statins in atherosclerotic disease: Focus on the antioxidant activity of atorvastatin. Curr Top Med Chem 2014;14:2542-51.
- Bang CN. Okin PM. Statin treatment, new-onset diabetes, and other adverse effects: A systematic review. Curr Cardiol Rep 2014;16:461.
- Wanders D, Plaisance EP, Judd RL. Pharmacological effects of lipid-lowering drugs on circulating adipokines. World J Diabetes 2010;1:116-28
- Ando H, Sugimoto K, Yanagihara H, et al. Effects of atorvastatin and pravastatin on glucose tolerance, adipokine levels and inflammatory markers in hypercholesterolaemic patients. Clin Exp Pharmacol Physiol 2008;35:1012-7.
- Von Eynatten M, Liu D, Bluemm A, et al. Changes in adiponectin multimer distribution in response to atorvastatin treatment in patients with type 2 diabetes. Clin Endocrinol (Oxf) 2009;71:27-32.
- American Diabetes Association. Standards of Medical Care in Diabetes-2012. Diabetes Care 2012;31:S11-S63.
- Barquera S, Campos-Nonato I, Hernández-Barrera L, et al. Prevalencia de obesidad en adultos mexicanos, 2000-2012. Salud Pública Méx 2013;55(Suppl 2):S151-S160.
- Mahadik SR, Lel RD, Mehtalia SD, Deo SS, Parikh V. Secretion of adiponectin from human subcutaneous and omental adipose tissue: Effects of ramipril and TNFα. Int J Diabetes Dev Ctries 2015;35:143-8.
- Sadowitz B, Maier KG, Gahtan V. Basic science review: Statintherapy – part I: The pleiotropic effects of statins in cardiovascular disease. Vasc Endovascular Surg 2010;44:241-51.
- von Eynatten M, Schneider JG, Hadziselimovic S, et als. Adipocytokines as a novel target for the anti-inflammatory effect of atorvastatin in patients with type 2 diabetes. Diabetes Care 2005;28:754-5.
- *Corresponding Author:
- Dr Alberto Francisco Rubio-Guerra
Motozintla # 30, Col Letran valle, México, DF CP 03600
Tel: 52-555-539-3584
Fax: 52-555-539-3584
E-mail: clinhta@hotmail.com
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http:// creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
BACKGROUND: Adipocytokines are adipocyte-secreted hormones that have a role in the effects of insulin, metabolism, endothelial function, vascular damage and cardiac disease. Statins have been shown to have cardioprotective effects and to modulate adipose tissue function; however, the effect of each statin on the circulating concentrations of adipocytokines varies.
OBJECTIVES: To evaluate the effect of atorvastatin on adipocytokine levels in normotensive patients with type 2 diabetes.
Methods: Twenty patients with type 2 diabetes and naive to statins and inhibitors of the renin-angiotensin systems received 10 mg atorvastatin once per day over a three-month period. Adiponectin and resistin levels were measured using ELISA at the beginning and conclusion of the study. Patients were evaluated monthly for blood pressure, fasting serum glucose levels, lipid profile and adverse events. Statistical analysis was performed using the Mann-Whitney U test
Results: All patients demonstrated a significant reduction in lipid values. Atorvastatin modestly increased the levels of adiponectin (6.85 μg/mL to 8.43 μg/mL; P=0.0602) and significantly decreased the levels of resistin (15.65 pg/mL to 10.09 pg/mL; P=0.022). None of the patients experienced adverse events.
Conclusion: Our results demonstrate that atorvastatin significantly reduced the levels of resistin and induced a modest increase in adiponectin levels. This may, in part, explain the cardiovascular effects observed with statins in diabetic patients.
-Keywords
Adiponectin; Atorvastatin; Diabetic patients; Resistin
Adipose tissue has been previously described as an active endocrine organ, and adiponectin and resistin have been described as peptides that are secreted by adipocytes (adipocytokines). Both hormones modulate the effects of insulin. In addition, hypoadiponectinemia and hyper-resistinemia are associated with hypertension, obesity and type 2 diabetes [1]. It has also been reported that hypoadiponectinemia is a risk factor in the development of cardiovascular disease [2] and hyperresistinemia is associated with inflammation and the pathways that lead to atherosclerosis [3]. Rubio et al [1] demonstrated that the adiponectin/resistin index correlates with carotid intima-media thickness.
Statins are effective cholesterol-lowering drugs that reduce the risk for stroke and cardiovascular disease, especially in diabetic patients [4]. However, beyond their cholesterol-lowering effects, statins have lipid-independent (or pleiotropic) properties, including reduction of oxidative stress, normalization of endothelial function and antiinflammatory actions [5].
Unfortunately, the use of statins includes the potential risk for adverse effects, such as myopathy, rhabdomyolysis and increasing levels of transaminases. In addition, recent evidence suggests an increased risk for new-onset diabetes mellitus [6].
Several pharmacological agents affect the production of adipocytokines. Statins modulate the production of adipocytokines and may modify insulin sensitivity as a consequence of an increase or reduction in circulating adipocytokines [7]. In fact, pravastatin and pitavastatin increase serum adiponectin concentrations, whereas simvastatin decreases its circulating levels.
Contradictory results have been reported regarding the effect of atorvastatin on adiponectin levels. Some authors have found that atorvastatin increases the levels of this adipocytokine [8], whereas other studies have reported no change in total adiponectin in response to atorvastatin [9]. Most studies have found that statin therapy has little to no effect on circulating resistin levels [7].
The aim of the present study was to evaluate the effect of atorvastatin on adipocytokine levels in normotensive patients with type 2 diabetes in Mexico.
Methods
A total of 20 normotensive patients with type 2 diabetes mellitus of >12 months’ duration and without previous treatment with thiazolindinediones, statins or inhibitors of the renin-angiotensin system were included. All patients received 10 mg atorvastatin daily, for the duration of the three-month study.
Diagnosis of type 2 diabetes was made according to the American Diabetes Association criteria [10].
Venous samples were collected in the morning after a 12 h overnight fast at the beginning and the conclusion of the study for all patients. Duplicate samples of circulating levels of adiponectin and resistin were measured using ELISA (R&D Systems, USA) according to the manufacturer’s recommendations. ELISAs were performed by personnel blinded to the study. Intra-assay coefficient of variability (CV) for adiponectin was 3.4% and interassay CV was 5.8%. Whereas intra-assay CV for resistin was 5.5% and inter-assay CV was 9.2%. Fasting glycemia and glycated hemoglobin (HbA1c) levels were measured, and lipid profiles were recorded.
Patients with any of the following diagnoses were excluded from the study: heart, hepatic or renal failure; evidence of valvular heart disease; heart block or cardiac arrhythmia; acute coronary syndrome or cerebrovascular disease diagnosed six months before the study’s initiation; autoimmune disease; pregnancy; urinary tract infection; fever; or a history of alcohol abuse and/or psychotropic drugs.
The study was conducted with the approval of the research and medical ethics committee of the authors’ hospital, in accordance with the Declaration of Helsinki. Participants provided written informed consent before their inclusion in the study protocol.
Statistical analysis
Data are presented as the mean ± SD. Statistical analysis was performed using the Mann-Whitney U test; P<0.05 was considered to be statistically significant.
Results
Basline characteristics of the patients are presented in Table 1. For all patients, mean (± SD) low-density lipoprotein (LDL) levels were reduced (4.013±0.46 mmol/L to 2.72±0.32 mmol/L; P<0.01).
| Age, years | 54±9.2 |
| Sex, male/female, n/n | 8/12 |
| Glycemia, mmol/L | 7.54±1.48 |
| Glycated hemoglobin, % | 6 |
| Low-density lipoprotein, mmol/L | 4.013±0.46 |
| Body mass index, kg/m2 | 32.4±5.1 |
| Blood pressure, systolic/diastolic, mmHg | 122/72 |
| History of diabetes mellitus, years | 8.8 |
Data presented as mean or mean ± SD unless otherwise indicated
Table 1: Baseline patient characteristics
Atorvastatin modestly increased the levels of adiponectin by 1.67 μg/mL (6.85 μg/mL to 8.43 μg/mL [95% CI 1.4 to 4.7]; P=0.0602), and significantly decreased the levels of resistin by 5.51 pg/mL (15.65 pg/mL to 10.09 pg/mL [95% CI 3.92 to 6.83]; P=0.022) (Figure 1). None of the patients experienced adverse events.
Discussion
The Mexican National Survey on Health and Nutrition 2012 reported that for Mexicans >20 years of age, 32.4% were affected by obesity and 38.8% were overweight, contributing to a high prevalence of type 2 diabetes and cardiovascular disease in Mexico [11]. Due to the role of hypoadiponectinemia and hyperresistinemia in the development of insulin resistance and in the pathways that lead to atherosclerosis, both adipocytokines are potential treatment targets. In fact, increasing adiponectin and reducing resistin levels contribute to reducing the risk for cardiovascular disease [1,12].
In the present study, we found that therapy with 10 mg atorvastatin daily for 12 weeks significantly reduced LDL and resistin levels in normotensive patients with type 2 diabetes, whereas statin induced a modest, but insignificant, increase in adiponectin. It is important to note that in our study, circulating levels of adipocytokines were measured in duplicate; therefore, intraindividual variation was taken into account when statistical analyses were performed.
It is also important to note that our patients were naive to thiazolindinedione, statin and angiotensin-converting enzymes/angiotensin II receptor blockers, because these drugs have been shown to modify circulating levels of adipocytokines [1].
Statins have been demonstrated to reduce cardiovascular risk in diabetic patients due to their lipid dependent [4] and lipid independent (or pleiotropic) actions [13]. In addition, their use in type 2 diabetic patients is effective and safe; in fact, the American Diabetes Association recommends that statins should be added, regardless of lipid value, to the treatment regimens of diabetic patients with overt cardiovascular disease, or in individuals without cardiovascular disease but who are >40 years of age and have ≥1 cardiovascular risk factor(s) [10].
Our results are consistent with those reported by Von Eynatten et al [9], who found no change in total adiponectin in response to the statin in diabetic patients [9]; however, Ando et al [8] found a significant increase in the adipocytokine levels in 36 hypercholesterolemic patients. It is important to note that in the study by Von Eynatten et al [9], and in our study, all patients were diabetic, whereas in the study by Ando et al [8], only four (11%) patients were diabetic. Studies comparing the effect of atorvastatin on circulating adiponectin levels in diabetic and nondiabetic patients are needed.
To our knowledge, the present study is the first to report a significant reduction in circulating resistin levels with the use of 10 mg of atorvastatin, whereas others reports have reported no change in resistin levels with that dose [7]. We do not have an explanation for this difference, although ethnicity may play a role. In fact, Von Eynatten et al [14] have shown that 40 mg of atorvastatin significantly reduces the circulating levels of resistin.
The increased incidence of diabetes and obesity in Mexico is accompanied by a greater incidence of cardiovascular complications, and the use of lipid-lowering drugs is accompanied not only by reductions in LDL levels, but also by a reduction in cardiovascular risk due to their pleiotropic effects. Our results demonstrate, in part, the pathways that lead to some of those pleiotropic actions.
Conclusion
Our results demonstrate that atorvastatin significantly reduced the levels of resistin and induced a modest, yet insignificant, increase in adiponectin levels. This may explain, in part, the lipid independent cardiovascular effects observed with statins in diabetic patients.
References
- Rubio-Guerra AF, Cabrera-Miranda JL, Vargas-Robles H, et al. Correlation between levels of circulating adipokines and adiponectin/resistin index with carotid intima-media thickness in hypertensive type 2 diabetic patients. Cardiology 2013;125:150-3.
- Behre CJ. Adiponectin, obesity and atherosclerosis. Scand J Clin Lab Invest 2007;67:449-58.
- Lozano-Nuevo JJ, Estrada-Garcia T, Vargas-Robles H, et al. Correlation between circulating adhesion molecules and resistin levels in hypertensive type-2 diabetic patients. Inflamm Allergy Drug Targets 2011;10:27-31.
- Colhoun HM, Betteridge DJ, Durrington PN, et al. CARDS investigators. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): Multicentre randomised placebo-controlled trial. Lancet 2004;364:685-96.
- Profumo E, Buttari B, Saso L, Rigano R. Pleiotropic effects of statins in atherosclerotic disease: Focus on the antioxidant activity of atorvastatin. Curr Top Med Chem 2014;14:2542-51.
- Bang CN. Okin PM. Statin treatment, new-onset diabetes, and other adverse effects: A systematic review. Curr Cardiol Rep 2014;16:461.
- Wanders D, Plaisance EP, Judd RL. Pharmacological effects of lipid-lowering drugs on circulating adipokines. World J Diabetes 2010;1:116-28
- Ando H, Sugimoto K, Yanagihara H, et al. Effects of atorvastatin and pravastatin on glucose tolerance, adipokine levels and inflammatory markers in hypercholesterolaemic patients. Clin Exp Pharmacol Physiol 2008;35:1012-7.
- Von Eynatten M, Liu D, Bluemm A, et al. Changes in adiponectin multimer distribution in response to atorvastatin treatment in patients with type 2 diabetes. Clin Endocrinol (Oxf) 2009;71:27-32.
- American Diabetes Association. Standards of Medical Care in Diabetes-2012. Diabetes Care 2012;31:S11-S63.
- Barquera S, Campos-Nonato I, Hernández-Barrera L, et al. Prevalencia de obesidad en adultos mexicanos, 2000-2012. Salud Pública Méx 2013;55(Suppl 2):S151-S160.
- Mahadik SR, Lel RD, Mehtalia SD, Deo SS, Parikh V. Secretion of adiponectin from human subcutaneous and omental adipose tissue: Effects of ramipril and TNFα. Int J Diabetes Dev Ctries 2015;35:143-8.
- Sadowitz B, Maier KG, Gahtan V. Basic science review: Statintherapy – part I: The pleiotropic effects of statins in cardiovascular disease. Vasc Endovascular Surg 2010;44:241-51.
- von Eynatten M, Schneider JG, Hadziselimovic S, et als. Adipocytokines as a novel target for the anti-inflammatory effect of atorvastatin in patients with type 2 diabetes. Diabetes Care 2005;28:754-5.