Identification of an aberrant testicular vein draining the right kidney
Received: 22-Dec-2017 Accepted Date: Jan 12, 2018; Published: 29-Jan-2018, DOI: 10.37532/1308-4038.18.11.15
Citation: Kulesza R, Labranche L, Sweeney S, et al. Identification of an aberrant testicular vein draining the right kidney. Int J Anat Var. 2018;11(1):015-017.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Summary
The Venous drainage of the testicles is asymmetric. The left testicular vein drains into the left renal vein while the right vein drains into the inferior vena cava (IVC). However, this textbook pattern is commonly complicated by anatomical variations in the number of veins draining each testicle and their sites of termination. Herein, we describe a specimen with three right testicular veins. The medial two veins terminated by joining the IVC. The third, lateral testicular vein coursed superiorly and laterally, received a tributary from the lateral aspect of the right kidney, and then joined the right subcostal vein. Histological examination of the vein emerging from the lateral aspect of the kidney revealed this vessel penetrated beyond the renal capsule into the renal cortex. Such a variation appears to be extremely rare, but has clinical application in surgical approaches and might provide an additional route for metastasis of testicular cancer.
Keywords
Variant; Renal; Subcardinal vein
Introduction
Development of mature venous networks that serve the abdominal wall, kidneys, and gonads are complex and intimately intertwined. During embryogenesis there are a number of symmetric, longitudinally running venous channels that develop to drain these regions, including posterior cardinal, supracardinal, subcardinal, and azygos venous networks. The azygos network drains the body wall and forms the azygos system. The supracardinal network forms the infrarenal segment of the inferior vena cava (IVC) and the subcardial network forms the renal segment of the IVC that includes suprarenal veins, renal veins and gonadal veins [1].
Venous channels draining the testes emerge from the posterior and superior aspect of the gland and become the pampiniform plexus. This plexus of veins is contained within the spermatic cord where the plexus consists of 3 or 4 veins, traverses the inguinal canal and courses along the posterior abdominal wall, where the veins converge to form a single testicular vein. The right and left testicular veins terminate asymmetrically. The right testicular vein empties directly into the IVC just below the entry of the renal veins, while the left empties into the left renal vein [1]. The testicular veins typically include valves [1]. A detailed investigation of testicular veins revealed that these vessels in fact receive small tributaries from the renal capsule, ureter and colon [2]. Regardless, this textbook pattern of venous drainage is frequently complicated by duplications and abnormal sites of termination [3,4]. Notably, duplications of testicular veins are up to six times more common on the left side [4]. Herein, we aim to describe a rare variant of an accessory right testicular vein that receives a tributary from the lateral aspect of the kidney and terminates in the subcostal vein.
Case Report
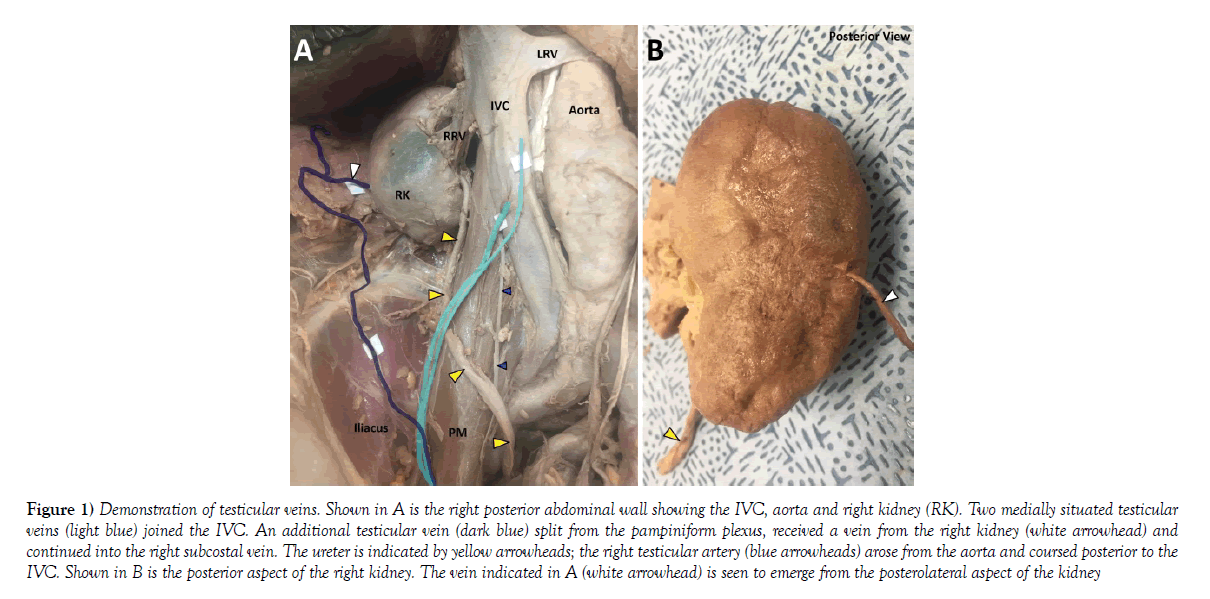
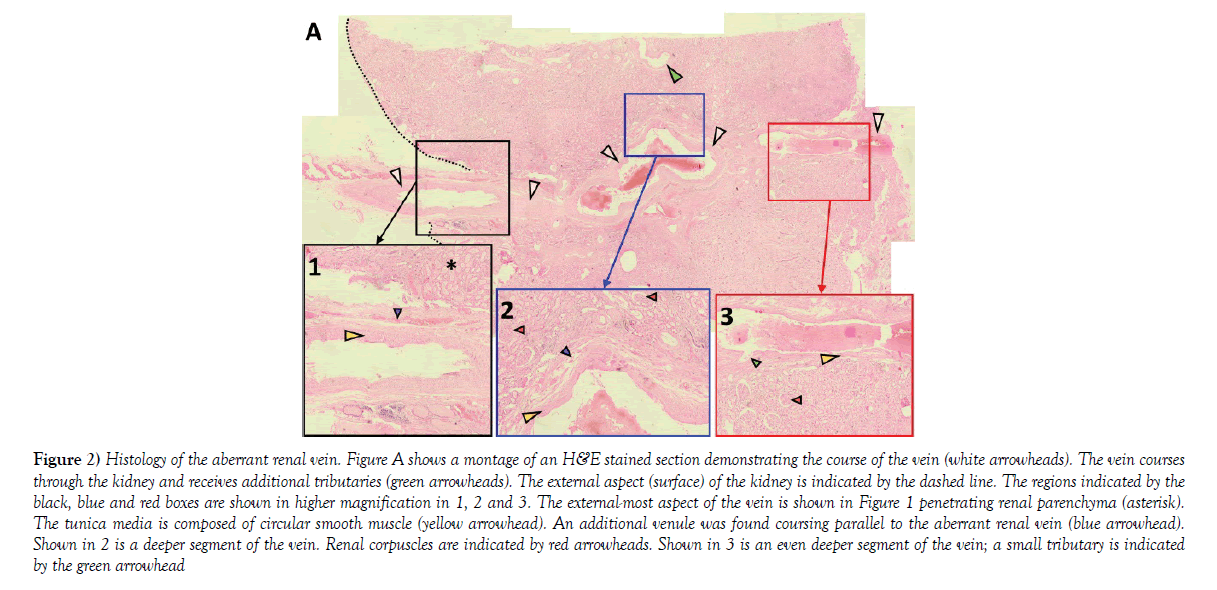
The cadaveric specimen described below was embalmed with Maryland State Blend embalming fluid at the Lake Erie College of Osteopathic Medicine and dissected under IRB exempt status. During routine dissection of the posterior abdominal wall of an 84-year-old male, three right testicular veins were identified (Figure 1A). Two of the veins coursed superior and medial to terminate by emptying into the anterior aspect of the IVC (Figure 1A, light blue). A third testicular vein coursed superiorly, anterior to the iliacus muscle and iliac crest, and crossed the ilioinguinal nerve. This aberrant testicular vein continued towards the pararenal fat, where it received a tributary from the posterolateral aspect of the right kidney (Figure 1A, white arrow). This vein then continued superiorly and joined the right subcostal vein. This aberrant testicular vein did not contain any valves along its course. Gross inspection of the right kidney after removal of the renal capsule revealed that the vein penetrated the renal cortex (Figure 1B). A block of tissue including the vein was dissected from the kidney, processed for routine histology and stained with hematoxylin and eosin. Histological examination of the vein emerging from the lateral aspect of the kidney confirmed that this vessel extended well past the renal capsule, penetrating deep into the renal parenchyma. A tissue section containing the vessel throughout the depth of the dissected block was reconstructed and is shown in Figure 2A. Tributaries of this vein are indicated by the green arrowheads. Segments of the medium-sized vein are shown at higher magnification in 1, 2 and 3. Figure 2.1 reveals that the tunica media of the vein is composed of circular smooth muscle (yellow arrowhead). Additionally, a small venule was found to course parallel to the renal vein (blue arrowheads in 1 and 2). Figure 2.2 demonstrates the vein coursing through the renal parenchyma; renal corpuscles are indicated by red arrowheads. Figure 2.3 reveals that the tunica media has been reduced to only a few layers of smooth muscle. No additional gross variants were identified in this cadaver.
Figure 1) Demonstration of testicular veins. Shown in A is the right posterior abdominal wall showing the IVC, aorta and right kidney (RK). Two medially situated testicular veins (light blue) joined the IVC. An additional testicular vein (dark blue) split from the pampiniform plexus, received a vein from the right kidney (white arrowhead) and continued into the right subcostal vein. The ureter is indicated by yellow arrowheads; the right testicular artery (blue arrowheads) arose from the aorta and coursed posterior to the IVC. Shown in B is the posterior aspect of the right kidney. The vein indicated in A (white arrowhead) is seen to emerge from the posterolateral aspect of the kidney
Figure 2) Histology of the aberrant renal vein. Figure A shows a montage of an H&E stained section demonstrating the course of the vein (white arrowheads). The vein courses through the kidney and receives additional tributaries (green arrowheads). The external aspect (surface) of the kidney is indicated by the dashed line. The regions indicated by the black, blue and red boxes are shown in higher magnification in 1, 2 and 3. The external-most aspect of the vein is shown in Figure 1 penetrating renal parenchyma (asterisk). The tunica media is composed of circular smooth muscle (yellow arrowhead). An additional venule was found coursing parallel to the aberrant renal vein (blue arrowhead). Shown in 2 is a deeper segment of the vein. Renal corpuscles are indicated by red arrowheads. Shown in 3 is an even deeper segment of the vein; a small tributary is indicated by the green arrowhead
Discussion
Variations in the number and drainage patterns of testicular veins are common. In fact, up to 21% of subjects have some degree of variation in normal testicular venous drainage, and such variants are significantly more common on the left side [3]. Furthermore, in a study of 120 gonadal veins, duplication of the right testicular vein was observed in only 5% of cases but duplication was observed in 30% of left testicular veins [4]. Additionally, right testicular veins terminate in the right renal vein in 10% of subjects [4]. There are only a few reports of more unusual variations of the testicular veins. Mazengenya described a specimen with four right testicular veins; one terminated by joining the IVC, two joined the right renal vein, and one appeared to terminate in the subcostal vein [5]. Gladstone described a specimen with a right ovarian vein that emptied into the IVC, but received an accessory renal vein from the inferior pole of the kidney, and a small tributary from the retroperitoneal adipose inferior to the right kidney [6]. Tubbs and coworkers presented a specimen where both right and left testicular veins received tributaries from the renal capsule, and then emptied into the subcostal vein [7]. The venous drainage pattern that we have described appears to be an extremely rare variant. The variation consists of a venous branch from the right pampiniform plexus which empties into the right renal vein via passage through the renal parynchyma and an additional branch which drains into the right subcostal vein. Altogether, this vein allows connections between the right testicle, right renal vein and the azygos system. Based on the development of the venous networks draining these structures, the venous channel described herein appears to be derived from a persistent subcardinal vein and a supracardinal-subcardinal anastomosis that was entrapped by the developing metanephros.
A thorough understanding of anatomical variation, regardless of how infrequent, is essential for accurate interpretation of radiological imaging and surgical treatment of patients. Not surprisingly, it is estimated that a significant number of malpractice cases are linked to inadequate knowledge of anatomical variation [8]. The anatomical variant described here can have clinical significance because of the structures/veins with which it communicates and its anatomic location. This vein would be at risk of injury during abdominal procedures (e.g. retroperitoneal lymph node dissection) [9] or could be inadvertently ligated, resulting in ischemic injury to the kidney or testicle [10]. Because the left testicular vein empties into the renal vein, compression of the left renal vein can lead to varicocele. As such, the vast majority of varicoceles occur on the left side of the body. However, when varicoceles do occur on the right side, they are typically associated with abdominal neoplasia [11]. Furthermore, duplicated testicular veins are a known risk factor for recurrence of varicocele following retroperitoneal approaches [12]. Because of the size and location of the aberrant renal vein, it could be mistaken for renal carcinoma, a renal cyst or a bifid or ectopic ureter on an abdominal CT. Finally, spread of testicular cancer is most commonly through lymphatic channels, but there are reported cases of metastasis to the kidney [13,14]. While metastasis to the kidney via lymphatic channels is possible, a venous metastatic route without valves may be a more likely pathway. The variant identified here could provide such a venous route, because of the lack of valves, for metastasis of testicular cancer to the ipsilateral kidney or for renal carcinoma to spread to the ipsilateral testicle.
Acknowledgement
The authors would like to thank Dr. Lisa Stevens for assistance with histology and the donor and his family for their generous donation and contribution to medical education.
REFERENCES
- Standring S. Gray’s Anatomy: The anatomical basis of clinical practice. 2008;1219-20.
- Wishahi MM. Anatomy of the venous drainage of the human testis: testicular vein cast, microdissection and radiographic demonstration. A new anatomical concept. Eur Urol. 1991;20:154-60.
- Asala S, Chaudhary SC, Masumbuko-Kahamba N, et al. Anatomical variations in the human testicular blood vessels. Ann Anat. 2001;183:545-9.
- Gupta R, Gupta A, Aggarwal N. Variations of gonadal veins: embryological prospective and clinical significance. J Clin Diagn Res. 2015;9:8-10.
- Mazengenya P. Multiple variations of the renal and testicular vessels: possible embryological basis and clinical importance. Surg Radiol Anat. 2016;38:729-33.
- Gladstone RJ. Development of the Inferior Vena Cava in the Light of Recent Research, with Especial Reference to Certain Abnormalities, and Current Descriptions of the Ascending Lumbar and Azygos Veins. J Anat. 1929;64:70-93.
- Tubbs RS, Salter EG, Oakes WJ. Unusual drainage of the testicular veins. Clin Anat. 2005;18:536-9.
- Sanudo J, Vazquez R, Puerta J. Meaning and clinical interest of the anatomical variations in the 21stcentury. Eur J Anat. 2003;7:1-3.
- Castillo OA, Sanchez-Salas R, Alvarez JM, et al. Inferior vena cava anomalies during laparoscopic retroperitoneal lymph node dissection. J Endourol. 2008;22:327-31.
- Satyapal KS, Haffejee AA, Singh B, et al. Additional renal arteries: incidence and morphometry. Surg Radiol Anat. 2001;23:33-8.
- James William Ryan, Gavin Sugrue, Sandra Graham, et al. A rare testicular vein anatomical variant contributes to right-sided varicocoele formation and leads to the diagnosis of renal cell carcinoma. BMJ Case Rep. 2017.
- Salsano G, Puccianti F, Barattini M, et al. A Rare Anatomical Variant of Spermatic Vein as Cause of Recurrence after Surgical Correction of Varicocele. Urology. 2016;90:15-16.
- Husband JE, Bellamy EA. Unusual thoraco abdominal sites of metastases in testicular tumors. AJR Am J Roentgenol. 1985;145:1165-71.
- Kramer ZB, Smith BD. Symptomatic renal metastasis from testicular cancer. Cancer. 1993;15;71:3979-80.