The investigation of anti-bacterial activity of Reactive Red-120
Received: 16-Feb-2018 Accepted Date: Feb 16, 2018; Published: 29-Mar-2018
Citation: Tazehkand MN. The investigation of anti-bacterial activity of Reactive Red-120. J Pharmacol Med Chem 2017;2(1):1-3.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Reactive dyes are resistant to natural biodegradation, due to the aromatic rings in their structure. Among dyes used in textile industry, Reactive Red- 120 (RR-120) is one of the frequently used dyes in textile industries and is a potential threat to the aquatic environment due to its poor biodegradability. The aim of this research is to analyze the antibacterial activity of Reactive Red-120 on E. coli and Bacillus subtilis with MIC, MBC and disc diffusion tests. In this study dilution test was used to finding the minimum inhibitory concentrations (MIC) and minimum bactericidal concentrations (MBC) of the Reactive red-120. For disc diffusion assay test substance were testet at 125 μg, 250 μg, and 500 μg concentrations. MIC values of Reactive red-120 against Bacillus subtilis is 100 In this study dilution test was used to finding the minimum inhibitory concentrations (MIC) and minimum bactericidal concentrations (MBC) of the Reactive red-120. For disc diffusion assay test substance were tested at 125, 250, and 500 μg concentrations. MIC values of Reactive red-120 against Bacillus subtilis is 100 μg/ml, and MBC values was 1200 μg/ml, MIC values of Reactive red-120 against E. coli is 50 μg/ml, and MBC values is 800 μg/ml. The result of this study showed that Reactive red- 120 has not antimicrobial activity against Bacillus subtilis and E. coli, because diameter of inhibition zone at all concentration was zero mm. So, it can be concluded that Reactive red-120 might not pose a potenti
Keywords
Reactive red-120; E. coli; Bacillus subtilis; Anti-bacterial activity.
DYe is a material that has an affinity to the substrate to which it is being applied. The dye is usually used in solution, and may need a mordant to remedy the fastness of the dye on the fiber [1]. The generality of natural dyes is reproduced from plants, fungi and lichens. Plant-based dyes such as woad, indigo and saffron were important trade in the economies of Asia and Europe [2]. Archaeological proofs showed that, particularly in India and Phoenicia, dyeing has been widely achieved for over 5,000 years. The detection of synthetic dyes late in the 19th century ended the large-scale market for natural dyes. The first human accomplished organic aniline dye, mauveine, was found serendipitously by William Henry Perkin in 1856, the result of a failed attempt at the total synthesis of quinine. Other aniline dyes followed, such as fuchsine, safranine, and induline. Many thousands of synthetic dyes have since been prepared [3,4]. Dyes are classified according to their used and chemical schema, and are composed of a group of atoms known as chromophores, responsible for the dye color. These chromophorecontaining centers are based on various functional groups, such as azo, anthraquinonoid, methine, nitro, aril methane, carbonyl and others. Also, electrons withdrawing or donating substituents so as to generate or intensify the color of the chromophores are denominated as auxo chromes [5-7].
Azo dyes are organic compounds including the functional group R-N=N-R′, in which R and R′ are generally aryl. They are a commercially important family of azo compounds containing the linkage C-N=N-C. Azo dyes are pervasively used to treat textiles, leather articles, and some foods [8,9]. Some classes of azo dyes include disperse Dyes, metal-complex dyes, reactive dyes and substantive dyes. Also called direct dyes, substantive dyes are used for cellulose-based textiles, which include cotton. The dyes bind to the textile by non-electrostatic forces. In another sorting, azo dyes can be classified according to the number of azo groups [10]. Many azo dyes are nontoxic, although some azo dyes such as dinitroaniline orange, ortho-nitroaniline orange, or pigment orange 1, 2, and 5 showed that mutagenic and carcinogenic effects on several test systems [11,12]. In a reactive dye, a chromophore consists of substituent that reacts with the substrate. Reactive dyes majority use in dyeing of cellulose same cotton or flax, but also wool is dying with reactive dyes. Reactive dyeing is the most significant method for the coloration of cellulosic fibres. Reactive dyes can also be applied on wool and nylon; in the latter case they are applied under weakly acidic conditions [13]. Reactive Red remains a popular platform for reactive dyes. Generally, reactive dyes are resistant to natural biodegradation, due to the aromatic rings in their structure [14]. Among dyes used in textile industry, Reactive Red-120 (RR- 120) is one of the frequently used dyes in textile industries and is a potential threat to the aquatic environment due to its poor biodegradability [15]. The aim of this research is to analyze the antibacterial activity of Reactive Red-120 on E. coli as a gram negative bacterial strain and Bacillus subtilis as a gram positive bacterial strain with MIC, MBC and disc diffusion tests.
Materials and Methods
Bacterial strains and culture conditions
The bacterial strains used as test organism were Escherichia coli as a gram negative bacterial strains and Bacillus subtils as gram positive bacterial strains obtained from Department of Biology of Pamukkale University. All bacteria were cultured aerobically at 37°C in LB agar and LB broth medium. Before experimental use, cultures from agar medium were sub cultivated in liquid media, incubated for 12 h (37°C).
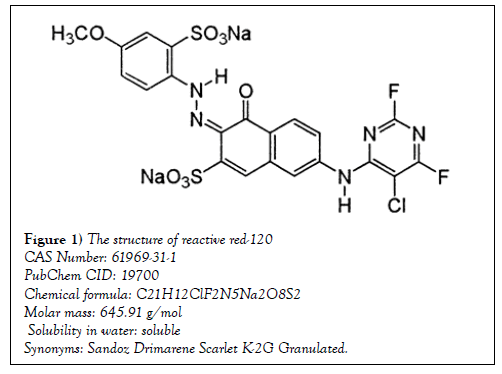
In this study, LB Broth, LB agar, Mueller Hinton broth and Mueller Hinton agar were purchased from Merck and the test substance, Reactive Red- 120 was purchased from Sigma-Aldrich and its properties and molecular structure is shown in Figures 1 and 2.
Antibacterial activity assay
McFarland Standards are used to standardize the proximate number of bacteria in a broth suspension by comparing the turbidity of the test suspension with that of the McFarland Standard. A McFarland Standard is a chemical solution of barium chloride and sulfuric acid; the reaction between these two chemicals results in the production of a fine precipitate, barium sulfate [16,17].
E. coli and Bacillus subtilis colonies were selected from solid medium using a sterile inoculation loop and inoculated into 10 ml of Mueller-Hinton broth in a sterile tube and mixed well. Turbidity levels of bacterial suspension were equated in accordance with the turbidity of 0.5 McFarland turbidity standards, which were prepared commercially. The accuracy of bacterial suspension was approved using a spectrophotometer analysis. Sterile Mueller- Hinton broth was used and measured at a wavelength of 625 nm as a blank solution for comparison [18].
Tube dilution test as described by Caburian and Osi with some modifications was used to finding the minimum inhibitory concentrations (MIC) and minimum bactericidal concentrations (MBC) of the Reactive red-120 against the E. coli and Bacillus subtilis [19]. Thirteen sterile glass tubes (12 ml) were numbered 1 to 13. Different concentrations of Reactive red-120 (final concentrations of 1.56 μg/ml to 6400 μg/ml) were added to different tubes with 4 ml Mueller Hinton broth. So, a microbial suspension of 0.5 McFarland of bacterial strains were cultured in Mueller Hinton broth containing different Reactive red- 120 concentrations tubes. The tubes were then incubated at 37°C for 12 hours. The tube with the lowest concentration (highest dilution) of the reactive red at which no visible growth or turbidity was observed was reported as the minimum inhibitory concentration of the oil on the test bacteria. The tubes were shaken to homogenize the contents and 150 μL of the contents of each tube was sub cultured by spreading Mueller-Hinton agar plastic petri dishes. The plates were incubated for 12 hours and then observed for any growth of colonies. Minimum bactericidal concentration was determined as the lowest concentration of Reactive red- 120 at which no colony observed following the sub culturing onto Mueller Hinton agar mediums [20].
For disc diffusion assay Reactive red-120 were dissolved in water until a concentration of 25 mg/ml was reached. Bacterial inoculum was set according to the McFarland 0.5 standard, and then 150 μl of bacterial suspension was taken using a sterile micropipette from each bacterial inoculum to be overlaid on the Mueller Hinton agar. İn this research reactive red-120 was tested at 125, 250, and 500 μg concentrations. Distilled water was used as negative control and each test set was repeated 3 times. Plates were incubated at 37°C and the inhibition zones were measured after 24 hours [21].
Results
The MIC and MBC of Reactive red-120 on Bacillus subtilis and E. coli were determined by dilution (1.56 μg/ml to 6400 μg/ml) method. The result of this study showed that MIC values of Reactive red-120 against Bacillus subtilis is 100 μg/ml, and MBC values was 1200 μg/ml, MIC values of Reactive red- 120 against E. coli is 50 μg/ml, and MBC values is 800 μg/ml.
In this study we observed that the E. coli (as a gram negative bacterial strain) was more sensitive to Reactive red-120 than Bacillus subtilis (as Gram-positive bacterial strains).
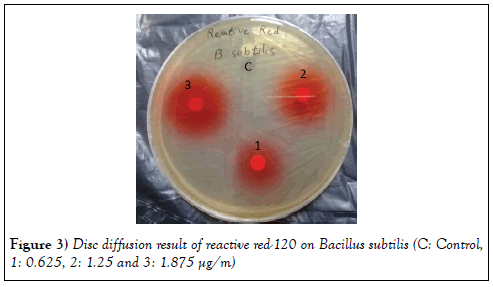
In our research, Reactive red-120 concentrations (125, 250, and 500 μg) were selected for disc diffusion assay. The disc diffusion values of Reactive red-120 against Bacillus subtilis and E. coli is shown in Figures 3 and 4. The result of this study showed that Reactive red-120 has not antimicrobial activity against Bacillus subtilis and E. coli, because diameter of inhibition zone at all concentration of Reactive red-120 against two bacterial strains was zero mm.
Figure 3: Disc diffusion result of reactive red-120 on Bacillus subtilis (C: Control, 1: 0.625, 2: 1.25 and 3: 1.875 µg/m)
Figure 4: Disc diffusion result of reactive red-120 on E. coli (C: Control, 1: 0.625, 2: 1.25 and 3: 1.875 µg/ ml)
Discussion
Azo dyes, a type of textile colorant, are integral to the textile industry and make up 70% of commercial dyes. Research has shown that some Azo dyes pose very serious health risks to humans if they are used in particular textiles and if they get into certain water supplies. The textile industry is a heavy polluter of waste gas, solids, water and noise. Wastewater is the most environmentally damaging, and the effluent from textile plants is classified as the most polluting of all the industrial sectors [22].
The results of this study showed that Reactive red-120 has not antibacterial effect on Bacillus subtilis and E. coli. According to our knowledge, it was the first study that addresses the antibacterial effects of Reactive red-120 against Bacillus subtilis and E. coli. We are using different dye in our life. Some of this dye is for food coloring and some of these are for textile industries. We are using textile and food dye in our life but some of dyes have toxic effects, for this reason we need to study about these substances. Chromosome aberration (CA) assay is one of methods to measure cytotoxicity and genotoxicity of a test substance [23]. For example, we studied cytotoxic, genotoxic and protective effects of a food coloring (4-MEI) with CA assay and another method, the results of these studies showed that 4-MEI have cytotoxic, anticancer and antioxidant effect but don’t have genotoxic and antigenotoxic effect on different test system [24-27]. There are many examples for textile dye that has toxic or antibacterial property against different bacterial strains. Leme and et al. reported that reactive green 19 textile dye has genotoxic effect on human dermal equivalent cells [28]. In another research the genotoxic effect of Disperse Red 1 was studied in mouse germ cells. The results obtain from this study was showed the harmful activity of this dye on reproductive health [29]. Similarly, antimicrobial effect of Quercus infectoria was studied on Escherichia coli, Bacillus subtilis, Klebsiella pneumoniae, Proteus vulgaris and Pseudomonas aeruginosa. The results obtained from this study showed that Quercus infectoria have antimicrobial activity on different bacterial strains [30]. In contrast to Reactive red-120, some of dyes have antimicrobial activity on gram positive and gram negative bacterial strains. MAJHI et al. observed that the dye obtained from Morinda citrifolia have antimicrobial activity on Klebsiella pneumoniae, Escherichia coli, Candida albicans and Aspergillus sp. [31]. Gul and Bakht reported that turmeric dye inhibited Escherichia coli and Staphylococcus aureus growth by disk diffusion analysis [32].
Conclusion
There was no report regarding to the antimicrobial effect of Reactive red- 120 in the available literatures. As our result, Reactive red-120 doesn’t have antibacterial effect on Bacillus subtilis and E. coli. So, it can be concluded that Reactive red-120 might not pose a potential risk for nature. However, it must be evaluated with different new studies.
REFERENCES
- Booth G, Zollinger H, McLaren K, et al. Dyes, general survey. Ullmann's Encyclopedia of Industrial Chemistry. 2000.
- Castillejo M, Moreno P, Oujja M, et al. editors. Lasers in the Conservation of Artworks: Proceedings of the International Conference Lacona VII, Madrid, Spain, 17-21 September 2007. CRC Press; 2008.
- Hunger K. Industrial dyes: Chemistry, properties, applications. John Wiley & Sons; 2007.
- Zollinger H. Color chemistry: Syntheses, properties, and applications of organic dyes and pigments. John Wiley & Sons; 2003.
- Christie RM. Colour chemistry. RSC Adv; 2001.
- Dos Santos AB, Cervantes FJ, Van Lier JB. Review paper on current technologies for decolourisation of textile wastewaters: Perspectives for anaerobic biotechnology. Bioresour Technol. 2007;98(12):2369-85.
- Prasad AA, Rao KB. Physico chemical characterization of textile effluent and screening for dye decolorizing bacteria. Global J Biochem Biotechnol.2010;5(2):80-6.
- TFL Leather Technology Ltd. European Ban on Certain Azo Dyes Archived. Dr. A. Quality and Environment, TFL. 2012.
- Hunger K, Mischke P, Rieper W, et al. Azo dyes in Ullmann’s Encyclopedia of Industrial Chemistry.
- Gilli P, Bertolasi V, Pretto L, et al. The nature of solid-state N− H⊙⊙⊙ O/O− H⊙⊙⊙ N tautomeric competition in resonant systems. Intramolecular proton transfer in low-barrier hydrogen bonds formed by the ⊙⊙⊙ OC − CN− NH⊙⊙⊙⇄⊙⊙⊙ HO− CC− NN⊙⊙⊙ Ketohydrazone − Azoenol System. A variable-temperature x-ray crystallographic and DFT computational study. J Am Chem Soc. 2002:124(45):13554-13567.
- Health & Safety in the Arts. A searchable database of health & safety information for artists. Tucson University Studies. Archived from the original on 2009.
- Engel E, Ulrich H, Vasold R, et al. Azo pigments and a basal cell carcinoma at the thumb. Dermatology. 2008;216(1):76-80.
- McNaught AD, McNaught AD. Compendium of chemical terminology. Oxford: Blackwell Science. 1997.
- Karacakaya P, Kılıc NK, Duygu E, et al. Stimulation of reactive dye removal by cyanobacteria in media containing triacontanol hormone. J Hazard Mater. 2009;172(2-3):1635-9.
- Paul J, Rawat KP, Sarma KSS, et al. Decoloration and degradation of Reactive Red-120 dye by electron beam irradiation in aqueous solution. Appl Radiat Isot. 2011; 69:982–7.
- Mcfarland standard -For in vitro use only- Catalogue No. DALYN Biologicals. TM50-TM60.
- McFarland J. Nephelometer: An instrument for media used for estimating the number of bacteria in suspensions used for calculating the opsonic index and for vaccines. J Am Med Assoc 1907;14:1176-8.
- Chamidah A, Hardoko A, Prihanto A. Antibacterial activities of β−glucan (laminaran) against gram-negative and gram-positive bacteria. AIP Conference Proceedings.2017:1844.
- Caburian AB, Osi MO. Characterization and evaluation of antimicrobial activity of the essential oil from the leaves of Piper beetle. EISRJC.2010;2(1):2-13.
- Guevara BQ. A guidebook to plant screening: Phytochemical and biological. University of Santo Tomas Publishing House; 2005.
- Souto R, Andrade AF, Uzeda M, et al. Prevalence of" non-oral" pathogenic bacteria in sub-gingival biofilm of subjects with chronic periodontitis. Braz J Microbiol. 2006;37(3):208-15.
- Chequer FM, De Oliveira GA, Ferraz ER, et al. Textile dyes: Dyeing process and environmental impact. In: Eco-friendly textile dyeing and finishing. InTech, 2013.
- Norizadeh Tazehkand M, Topaktas M. The in vitro genotoxic and cytotoxic effects of remeron on human peripheral blood lymphocytes. Drug Chem Toxicol. 2015;38(3):266-71.
- Norizadeh MT. Anti-proliferative effect of a food coloring on colon cancer cell line. Bratisl Lek Listy. 2017;118(5):265-8.
- Tazehkand MN, Hajipour O, Moridikia F, et al. Investigation of anti-carcinogenic and antioxidant effects of-methylimidazole. Bratisl Lek Listy. 2017;118(3):149-52.
- Tazehkand MN, Moridikia A, Hajipour O, et al. The effects of 4-MEI on cell proliferation, DNA breaking and DNA fragmentation. Bratisl Lek Listy. 2016;117(7):371-5.
- Tazehkand MN, Topaktas M, Yilmaz MB, et al. Delineating the antigenotoxic and anticytotoxic potentials of 4-methylimidazole against ethyl methanesulfonate toxicity in bone marrow cell of swiss albino mice. Bratisl Lek Listy. 2016;117(5):290-4.
- Leme DM, Primo FL, Gobo GG, et al. Genotoxicity assessment of reactive and disperse textile dyes using human dermal equivalent (3D cell culture system). J Toxicol Environ Health A. 2015;78(7):466-80.
- Fernandes FH, Bustos-Obregon E, Salvadori DM. Disperse Red 1 (textile dye) induces cytotoxic and genotoxic effects in mouse germ cells. Reprod Toxicol. 2015;53:75-81.
- Singh R, Jain A, Panwar S, et al. Antimicrobial activity of some natural dyes. Dyes Pigm. 2005;66(2):99-102.
- Majhi B, Satapathy KB, Mishra SK, et al. Studies on anti-microbial effect of natural dyes and pigments obtained from the leaf of Artocarpus heterophyllus Lam. and Tectona grandis Lf. Phytomedicine. 2016;7(4):396-401.
- Gul P, Bakht J. Anti-microbial activity of turmeric extract and its potential use in food industry. J Food Sci Technol. 2015;52(4):2272-9.