Extensive hyperostosis cranii interna involving multiple cranial bones in a 79-year-old woman
2 Department of Surgery, Uniformed Services University of the Health Sciences, USA, Email: guinevere.granite@usuhs.edu
Received: 20-Nov-2018 Accepted Date: Dec 17, 2018; Published: 26-Dec-2018
Citation: Couture AK, Silverman MB, Thomas JZ, et al. Extensive Hyperostosis cranil interna involving multiple cranial bones in a 79 year-old woman. Int J Anat Var. Dec 2018;11(4):147-150.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
A thickening of the frontal bones characterizes Hyperostosis frontalis interna. This condition is found typically postmortem in post-menopausal women who undergo cadaveric dissection. Live patients commonly are asymptomatic, but may experience increased intracranial pressure, headaches, vertigo, visual and personality changes. Previous case studies report variable amounts of thickness in ostoses, but most remain localized to the frontal bone and extradural space. In contrast, our 79 year-old female cadavers exhibited circumferential thickening of the skull with subdural involvement. We found multiple ostoses bilaterally throughout the calvaria, but primarily endocranially along the frontal and parietal bones. We believe that this case is unique in describing bone growth beneath the dura mater. Using measurement software, we found dural thickening to be approximately doubled while the skull circumference was consistent with normal measurements. Thus, the dura mater may play a role in the development of Hyperostosis frontalis interna in exceptional situations.
Keywords
Extensive hyperostosis frontalis interna; Extensive hyperostosis cranii interna; Intradural ostoses; Estrogen; Postmenopausal
Introduction
Hyperostosis frontalis interna (HFI) is the thickening of the inner table of the frontal bone of the skull. In historical samples and modern studies, investigators have found HFI most often in post-menopausal women during cadaveric dissection and in imaging studies [1-3]. Frequent comorbid conditions they found were diabetes and obesity. The frequency of HFI in cadavers and dry skull materials was almost identical, 12.5% and 12.3%, respectively, and severe HFI was found in female cadavers over 65 years-old (87.5%) [4]. Classically, the thickening associated with HFI does not cross the midline and typically only expands to the periosteal dura [5].
Morgagni in 1719 first described HFI [6]. Then, it was presumed to be a feature of other syndromic disorders: Morgagni, Stewart-Morel and Troell- Junet syndromes [7]. Now, well described in the literature as a disease process separate from any individual syndrome, our case leads us to suggest that there is still much to learn about how the disease can progress in an individual throughout life. Hershkovitz characterized and established nomenclature for intracranial ostoses, including classification of hyperostosis cranii [5]. HFI as a pattern of growth under these classifications is further sub-divided by morphologic appearance and extent within the skull [5]. Here we present a case of extensive HFI with several unique features, including circumferential and intradural ostoses.
Case Report
Background
This 79-year-old African-American female cadaver (listed cause of death pulmonary embolism) was obtained after donation from the Maryland State Anatomy Board and transferred to the Uniformed Services University of the Health Sciences for cadaveric dissection. Upon postmortem examination, she exhibited an obese body habitus, artificial left knee, and a history of appendectomy. Upon removal of the calvaria, we observed extensive HFI. Unfortunately, the brain was not salvageable for examination after embalming. We observed no abnormalities of the external aspect of the calvaria. According to the policies of the Maryland State Anatomy Board, we were provided no other medical or surgical history.
Methodology
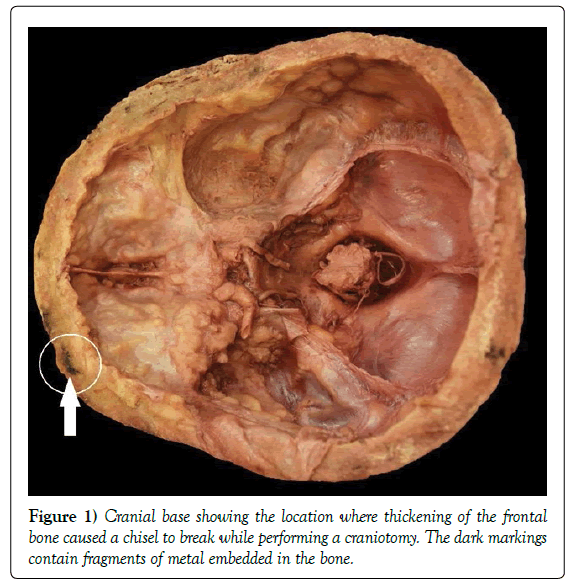
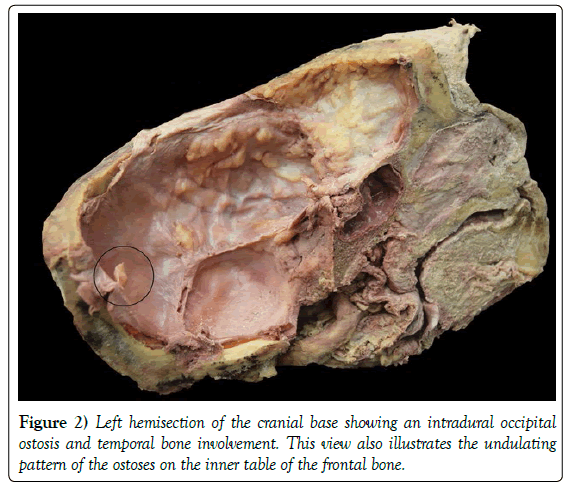
We discovered the presence and extent of the HFI during our dissection. We initially attempted a craniotomy using a Ridgid 12 V Job Max Power Base with a 1-1/8” Wood Plunge Cutting Blade and a Multi-Tool Tool Head Saw to dissect through the external bony lamina at the widest part of the skull circumferentially. The blade could not penetrate the full thickness of the skull. We then utilized Robox Stainless steel chisels and a Miltex Stainless steel post-mortem hammer to break through the internal bony lamina. As we began to utilize the chisel and hammer, the first chisel (0.5 inch blade width, 8 inch length) we used broke off into the frontal bone during the craniotomy. The region in which this chisel broke off is circled in Figure 1. Eventually, we successfully removed the calvaria. We encountered differences in the thickness of the frontal bone at the plane of the craniotomy between the calvaria and the cranial base, highlighting the undulation of the ostoses’ thickness (Figure 2). Unfortunately, due to the policies of the Maryland State Anatomy Board, the body was returned for cremation before we could obtain further measurements. Therefore, we calculated measurements of the skull and bony changes from photographic records using ImageJ, version 1.52a (National Institutes of Health, Maryland 2018) [8].
Calvaria
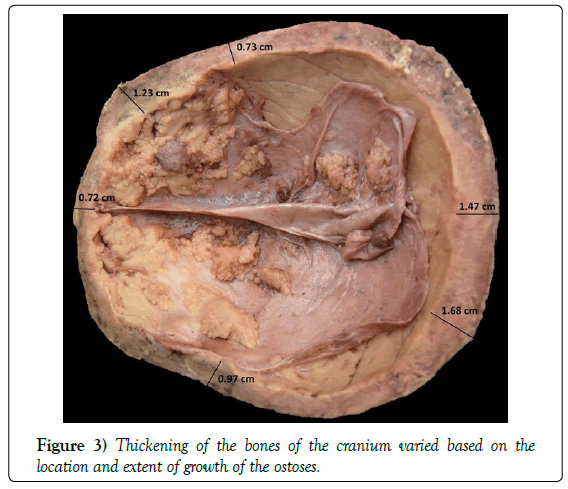
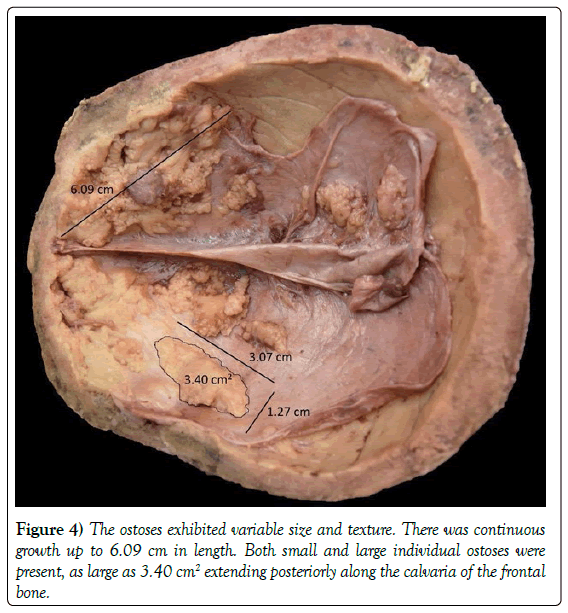
We found asymmetrical circumferential thickening of the inner table of the cranial bones in the plane of the craniotomy (Figure 3). Most noticeably, we observed the greatest thickness in the frontal and parietal bones (1.23 cm and 1.68 cm, respectively) (Table 1). On gross inspection of the calvaria, we found bilateral thickened bone and conjoined irregular ostoses. We observed a more significant thickening on the left frontal bone. We found significant ostoses within the dura of the calvaria. These particular intradural ostoses exhibited both continuous growth patterns and independent accumulations not connected to this outgrowth, extending toward the posterior portion of the dura. Their textures were also irregular, varying from smooth to rough. The ostoses were bilateral and displaced away from the midline (Figure 4).
| Location | Size (cm) | Normal Based on Mahind and Murty (cm) [21] | % of normal (%) |
|---|---|---|---|
| Frontal bone (midline) | 0.72 | 0.74 | 97.3 |
| Frontal bone (left lateral) | 1.23 | 0.58 | 212.07 |
| Left parietal bone | 0.73 | 0.56 | 130.36 |
| Right parietal bone | 0.97 | 0.57 | 170.18 |
| Occipital bone (midline) | 1.47 | 0.83 | 177.11 |
| Occipital bone (right lateral) | 1.68 | 0.73 | 230.14 |
Table 1: Selected measurements of thickness of the cranium in the plane of craniotomy1 , 1 Postmortem examination of females at University Malaya, Kuala Lumpur, Malaysia, 2009 [21].
Cranial base
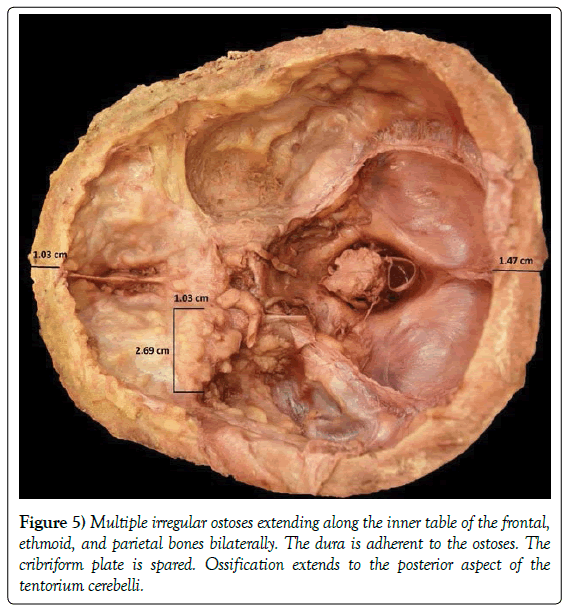
The most significant ostoses occurred on the frontal bone where the bones had fused visibly together in an undulating pattern. They extended to the ethmoid, sphenoid, temporal and parietal bones in the cranial base. The base of the left sphenoid bone was more involved (Figure 5). The cribriform plate was impinged posteriorly by the ostoses but is intact. There was also a single intradural ostosis on the occipital bone (Figure 2). The natural contours of the skull were asymmetric due to the endocranial ostoses (Figure 1).
There is significant compromise of the temporal lobe space on the left side, and very unusual temporal lobe contour on the right (Figure 1). While structures beneath the tentorium cerebelli are symmetric, the basilar artery is markedly dilated, and the posterior cerebellar space is compressed. There is also asymmetry of the internal carotid arteries.
Discussion
The incidence of HFI in cadaveric populations is approximately 4%-5% [9]. Multiple risk factors exist for its development [10]. While HFI is largely sex and age-dependent (female and advanced age), diabetes and obesity are also confounding factors [9-12]. Various other conditions, such as myotonic dystrophy and Morgagni’s syndrome also exhibit HFI [13]. HFI also associates with the diet and lifestyle of the modern era when compared with its prevalence in archeological specimens [1,11,14,15]. Since neuropsychiatric symptoms secondary to frontal lobe compression also associate with HFI historically, Parkinson’s disease is a frequent diagnosis [16]. In the modern era, these types of symptoms indicate the degree of compression and may also suggest the beginning of a degenerative process in the brain [17,18]. Many patients with extensive HFI exhibit neuropsychiatric symptomatology, ranging from depression to forgetfulness and impairment of working memory [16]. The extent of HFI present in this cadaver allows us to investigate further the disease process and its progression throughout life [19].
Hershkovitz et al. originally classified the bony growth of HFI by varying degrees of thickness of the frontal bone [5]. They described isolated ostoses occupying increasing amounts of the frontal bone (A-D), with D being the most extensive and indicative of continuous growth. Such severity involved compression of the underlying dura mater and brain tissue. However, Talarico et al. described the growth as limited to the frontal bones, extradural, and bounded posteriorly by the groove of the middle meningeal artery [6,20]. The cadaver we studied by contrast exhibits extensive hyperostosis, both circumferentially and intradurally. The growths we observed also crowded around the midline of the cranial base, with potential midline involvement at the lesser wing of the sphenoid.
When we measured calvarial thickness, we found it varied [21,22]. We found the thickness greatest on the right occipital bone (1.68 cm, 230.14% of the norm). Coincidently, we found the midline of the frontal bone, most commonly the thickest area of growth in HFI, was 0.72 cm (97.30% of the norm), providing further evidence for midline sparing of this pattern of ostoses on the frontal bone in this cadaver. The left frontal bone measured 1.23 cm (212.07% of the norm), a characteristic pattern of frontal bone thickening in HFI. Unexpectedly, we found the occipital bone was thicker than the frontal bone compared to that typically seen. Thus, we observed a circumferential, rather than predominantly frontal growth pattern. Additionally, we found the circumference of her calvaria approximated 46.15 cm, consistent with the normal range for her age group. This may imply that all of her bony growth took place intracranially, which may have put significant pressure on her brain, and thus may have led to symptoms during her life.
Raikos et al. reported a case with intradural ostoses consistent with the bone composition of HFI perforating the falx cerebri [4]. Intradural growths are not included in the initial A-D typing method outlined by Hershkovitz et al. [5]. Raikos et al. designated these intradural growths as “Type E” [4]. Our examination found far more extensive “Type E” growths along the dura mater on the frontal bone with two defined ostoses at the posterior parietal bone. These intradural ostoses were more extensive and continuous anterior to the coronal suture. These intradural ostoses seem to follow the Thevoz European model of HFI. In this model, the dura is responsible for the bone growth. Thus, the intradural ostoses we observed may have been early stage bone growths. Hershkovitz et al. suggested that the growths actually follow a more global model where neovascularization of the lamellar bone by the dura is one aspect of the growth [5]. Others have observed that dural cells are multipotent in vivo. Osteoblastic proliferation from the dura supports the importance of the dura in HFI, and may explain growths both intra- and extradurally [23].
As mentioned previously, HFI occurs primarily in females, and is observed after significant hormonal imbalance, such as menopause [2,3,23]. Yet, excess estrogen, especially as exposure to estradiol, through medical treatment may also provoke the development of HFI [24]. While the condition antemortem is commonly asymptomatic, symptoms may include headaches and psychiatric issues as comorbidities [10,16]. Though these symptoms appear to be more of a mass effect, associated concurrent conditions also involving hormonal influence include atherosclerosis, diabetic nephropathy, osteoporosis, and obesity [25,26].
Our knowledge of this individual’s medical history is limited. She was obese and her listed cause of death was a pulmonary embolism. Obesity exacerbates hormonal imbalance by producing peripheral estrogens, increasing the conversion of androstenedione to estrone, and by decreasing the availability of sex hormone binding globulin, thereby increasing available estradiol [27]. Obesity and states of estrogen excess, such as postmenopausal hormone replacement therapy (HRT), can also increase circulating clotting factors and decrease antithrombin by a multifactorial process [28].
Multiple factors may lead to an underlying estrogen imbalance. HFI is currently being observed and documented in younger populations with a 2.5 fold increase in risk. May et al. suggested that this may correlate with fertility changes and hormone treatments as well as dietary changes. HRT may exacerbate the development of HFI. Women may be put on HRT to mitigate the symptoms of menopause [29] and men may receive hormonal therapy for prostate cancer [30]. Historically, castration has been associated with the development of HFI [31]. Diet, such as the intake of fatty foods and/or phytoestrogens, are also indicated [29]. In the 18th century, those who could afford higher calorie diets, had a higher incidence of HFI. Those with less economic resources now experience more exposure to high caloric diets through fast food; a phenomenon correlated with obesity and hyperostosis [32].
Based on our hypothesis of dural involvement and etiology, we propose the following additions to the Hershkovitz and Raikos classification systems [4,5]: Grade E1-Severe HFI with soft tissue expansion, Grade E2-Continuous ostoses involving over 50% of the frontal endocranium and expansion to other bones of the cranium, Grade F – The presence of both grade E1 and E2 ostoses (Tables 2 and 3). We suggest the name STAGWAC Extension Score for this expanded grading scale. The acronym STAGWAC is a combination of the authors’ different last names. Because the discovery of this case was postmortem, we cannot determine whether the order in the disease progression occurs first in expansion to other bones of the cranium or expansion to soft tissue.
| Location | Size (cm) | Normal based on De Boer et al. (cm) | % of normal (%) |
|---|---|---|---|
| Frontal Bone | 0.72 | 0.68 | 105.88 |
| Left Parietal Bone | 0.73 | 0.44 | 165.91 |
| Right Parietal Bone | 0.97 | 0.44 | 220.45 |
| Occipital Bone | 1.47 | 0.81 | 181.48 |
Table 2: Limited skull measurements of comparative population, Autopsy reports of black females in the Netherlands, 2014-2016 [22].
| Grade of Severity | Morphological Description |
|---|---|
| Type A | Single or multiple isolated bony elevations less than 10 mm in diameter located on the endocranial surface of the frontal bone |
| Type B | Nodular bony formations occupying less than 25% of the frontal bone |
| Type C | Nodular bony formations occupying up to 50% of the frontal bone |
| Type D | Continuous nodular bony overgrowth involving over 50% of the frontal endocranium |
| Type E1 (b,c) | Severe hyperostosis frontalis interna with soft tissue expansion and/or intradural nodules |
| Type E2 (c) | Severe hyperostosis frontalis interna with Continuous nodular bony overgrowth involving over 50% of the frontal endocranium and expansion to other bones of the cranium |
| Type F (c) | The presence of both grade E1 and E2 lesions |
Table 3: Classification of HFI (a) HFI classification by Hershkovitz et al. (1999) [4], (b) Modification by Raikos et al. [5] Proposed modifications - STAGWAC Score.
Pathogenesis and its mechanism in HFI have been discussed at length in the literature. Although several mechanisms have been proposed, no definitive consensus exists as to the cause of HFI [4-6,23,29,30]. Since the history of this cadaver was minimal, we are unable to comment on diet or genetics as potential causes. Our goal is to add an interesting and unique case that may further the understanding of HFI progression.
Our findings of intradural ostoses support the speculation by Djonic et al. 2016 that the bone growth is dependent on the dura [23]. We propose that the dura may be involved in rare cases of HFI. In such cases, ostoses may arise by two processes. One is remodeling in the inner table of the calvaria. The second mechanism is the production of new bone via osteoblastic proliferation from the multipotent cells of the dura.
Conclusion
Our observation of the global extent of independent intradural ostoses in this 79-year-old female cadaver, leads us to suggest an expansion of the Hershkovitz and Raikos classification systems is warranted. We suggest the name STAGWAC Extension Score for the expanded grading scale we propose. In addition, the midline of the cranial base is characteristically spared in HFI. Thus, we believe our case exceeds the criteria of the Hershkovitz and Raikos classification systems. To our knowledge, previous case reports have not described this extent of hyperostosis and thus, ours may be a unique finding.
Acknowledgements
The authors would like to thank the family of the donor and the Maryland State Anatomy Board for their beneficent contribution. Without their generosity, this article would not have been possible. The authors would also like to thank HMC Ronald Rivenburgh, USN, Ret., the Uniformed Services University Anatomical Curator, for his support in the completion of this study.
REFERENCES
- Barber G, Watt I, Rodgers J. A Comparison of radiological and palaeopathological diagnostic criteria for hyperostosis frontalis interna. Int J Osteoarchaeol. 1997;7:157-64.
- Nikolic S, Djonic D, Z̆ivkovic V, et al. Rate of occurrence, gross appearance, and age relation of hyperostosis frontalis interna in females: A prospective autopsy study. Am J Forensic Med Pathol. 2010;31:205-7.
- May H, Peled N, Dar G, et al. Hyperostosis frontalis interna: Criteria for sexing and aging a skeleton. Int J Legal Med. 2011;125:669-73.
- Raikos A, Paraskevas GK, Yusuf F, et al. Etiopathogenesis of hyperostosis frontalis interna: A mystery still. Ann Anat. 2011;193:453-8.
- Hershkovitz I, Greenwald C, Rothschild BM, et al. Hyperostosis frontalis interna: An anthropological perspective. Am J Phys Anthropol. 1999;109:303-25.
- Talarico EF, Prather AD, Hardt KD. A case of extensive hyperostosis frontalis interna in an 87-year-old female human cadaver. Clin Anat. 2008;21:259-68.
- Champion T, Cope JM. A severe case of hyperostosis frontalis interna and multiple comorbidities. Int J Anat Var. 2012;5:76-8.
- Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9:671-5.
- Chaljub G, Johnson III RF, Johnson Jr. RF, et al. Unusually exuberant hyperostosis frontalis interna: MRI. Neuroradiol. 1999;41:44-5.
- Suzuki K, Kokubun N, Okamura M, et al. Hyperostosis frontalis interna in myotonic dystrophy. Intern Med. 2017;56:2539-40.
- Western AG, Bekvalac JJ. Hyperostosis frontalis interna in female historic skeletal populations: Age, sex hormones and the impact of industrialization. Am J Phys Anthropol. 2017;162:501-15.
- She R, Szakacs J. Hyperostosis frontalis interna: Case report and review of literature. Ann Clin Lab Sci. 2004;34:206-8.
- Champion T, Cope JM. A severe case of hyperostosis frontalis interna and multiple comorbidities. Int J Anat Var. 2012;5:76-8.
- Bebel A, Golijewskaja A. A case of hyperostosis frontalis interna from deir el-bahari, Egypt. BNE. 2015;9:45-54.
- Greig DM. On intracranial osteophytes. Edinb Med J. 1928;35:237-260.
- Gilbert T, Ait S, Delphin F, et al. Frontal cortex dysfunction due to extensive hyperostosis frontalis interna. BMJ Case Rep. 2012;1-3.
- Royle NA, Hernandez MC, Maniega SM, et al. Influence of thickening of the inner skull table on intracranial volume measurement in older people. Magn Reson Imaging. 2013;31:918-22.
- Batun GC, Yuruyen M, Vatankulu B, et al. Hyperostosis frontalis interna presenting as depression and parkinsonism in an older woman. Psychogeriatrics. 2015;16:329-30.
- Mckinney AM. Normal variations in calvarial contour, irregular ossification/aeration, and inward/outward projections/protuberances. Atlas Norm Imag Var Brain Skull Craniocerv Vasc. 2017;887-922.
- James AW, Theologis AA, Brugmann SA, et al. Estrogen/estrogen receptor alpha signaling in mouse posterofrontal cranial suture fusion. PLoS One. 2009;4:1-16.
- Mahinda HAM, Murty OP. Variability in thickness of skull bones and sternum-an autopsy experience. Indian J Forensic Med Toxicol. 2009;26:26-31.
- De Boer HH, Van der Merwe AE, Soerdjbalie-Maikoe VV. Human cranial vault thickness in a contemporary sample of 1097 autopsy cases: relation to body weight, stature, age, sex and ancestry. Int J Legal Med. 2016;130:1371-7.
- Djonic D, Bracanovic D, Rakocevic Z, et al. Hyperostosis frontalis interna in postmenopausal women-possible relation to osteoporosis. Women Health. 2016;56:994-1007.
- Harding FE. Endocrinopathies associated with hyperostosis frontalis interna. Am J Med. 1949;6:329-35.
- Ramchandren S, Liebeskind DS. Headache in a patient with Klinefelter’s syndrome and hyperostosis frontalis interna. J Headache Pain. 2007;8:342-4.
- Attanasio F, Granziera S, Giantin V, et al. Full penetrance of morgagni-stewart-morel syndrome in a 75-year-old woman: Case report and review of the literature. J Clin Endocrinol Metab. 2013;98:453-7.
- Siiteri PK. Adipose tissue as a source of hormones. Am J Clin Nutr. 1987;45:277-82.
- Yang Y, Wang L, Wu Z. Frontal sinus osteoma accompanied by intracranial mucocele and local hyperostosis frontalis interna. World Neurosurg. 2018;113:94-5.
- May H, Peled N, Dar G, et al. Hyperostosis frontalis interna: What does it tell us about our health? Am J Hum Biol. 2011;23:392-7.
- May H, Peled N, Dar G, et al. Hyperostosis frontalis interna and androgen suppression. Anat Rec. 2010;293:1333-6.
- Belcastro MG, Todero A, Fornaciari G, et al. Hyperostosis frontalis interna (HFI) and castration: The case of the famous singer Farinelli (1705-1782). J Anat. 2011;219:632-637.
- Flohr S, Witzel C. Hyperostosis frontalis interna-A marker of social status? Evidence from the Bronze-Age “high society” of Qatna, Syria. HOMO J Comp Hum Biol. 2011;62:30-43.