Intraspinal ventral extradural pseudocyst from C2- Th9 with compression of spinal cord: Case report and review of literature
2 Department of Radiology, Clemens Hospital, Academic Hospital of Muenster University, Muenster, Germany
Received: 17-Jan-2018 Accepted Date: Jan 29, 2018; Published: 05-Feb-2018
Citation: Fistouris P, Darvish O, Calero S, et al. Intraspinal ventral extradural pseudocyst from C2- Th9 with compression of spinal cord: Case report and review of literature. J Neurol Clin Neurosci. 2018;2(1):20-23.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Intraspinal extradural spinal cysts are rare lesions, which can induce symptoms due to spinal cord compression or even CSF low pressure syndrome. They are regarded to be result of a trauma, post-surgical, or even congenital. Depended on size and location of the cysts, patients could be asymptomatic for a long time until an enlargement of the cyst. To best of our knowledge, there is no previous report in literature with a 50 cm long intraspinal ventral extradural cyst causing spinal cord compression. Below we present our experience, we present our operative results, and provide a review of literature.
Keywords
Intraspinal; Extradural; Cyst; Spinal cord compression
Introduction
Intraspinal pseudocysts are relatively uncommon lesions, which can induce CSF low pressure syndrome or even a spinal cord compression. They thought to be associated with a trauma or just spontaneous. Some theories have been proposed to explain the pathogenic mechanism, from which the check -valve seems to be the most widely accepted. Some patients are asymptomatic, but most of them present with symptoms due to spinal cord compression depended on size of cyst and location. Below we present a case of a 40-year-old patient with an almost 50 cm long intraspinal ventral extradural pseudocyst.
Case Report
Presentation and examination
A 40-year old female patient was admitted to our neurosurgical clinics after a MRI-scan, which showed an extended intraspinal ventral extradural cystic lesion from C2 to Th9. The patient was complaining about progressive weakness and numbness of the left side, predominantly of the left arm. Furthermore, she had been suffering from a CSF low pressure syndrome, with occasional orthostatic bilateral headaches, which were appeared more frequently in the last few months. Furthermore, these were often accompanied by nausea, unsteadiness or vertigo. At the age of 8 she had a car accident, without an obvious injury or any neurological deficits, so that she was treated conservative. At the time of admission to the hospital the patient looked well, with obvious difficulties by standing or walking alone (atactic gait on the left side). Muscle strength was intact, as well as tonus. There was no muscle spasm or deformity of spine. There was found a hypoesthesia from C5 dermatome downward only on the left side of body. Romberg test became markedly unsteady with closed eyes (loss of proprioception). Vibration sensation was present bilaterally. There was showed a dysmetria of hands and legs, typified by the overshoot. Slight increased reflexes on the left side. Babinski sign was negative on both sides. No evidence of a cauda syndrome.
Imaging findings
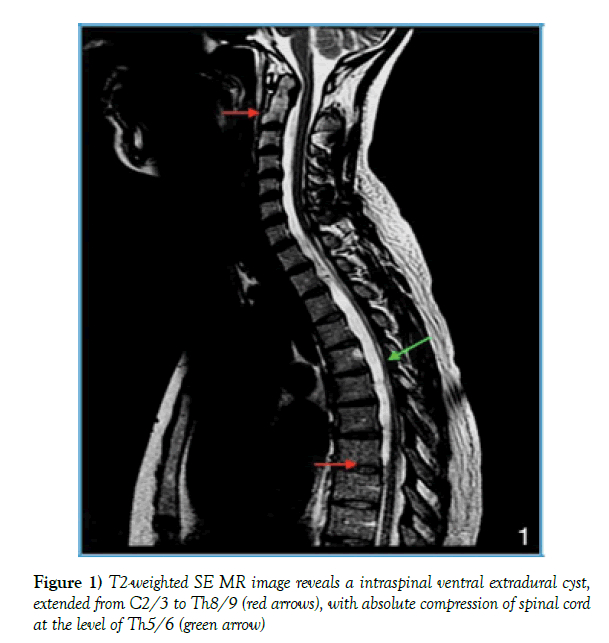
An external MRI of the cervical spine showed an incompletely imaged ventral longitudinal, intraspinal fluid collection (Figure 1). For further radiological presentation an ascending cervical myelography took place after stationary admission. The puncture was performed using a 22G needle (Braun) at level of L4/5 with intrathecal injection of 10 ml Imeron (Iomeprol) 300M.
In Trendelenburg position, contrast agent was found in the cervical dural sac, but not in the ventral cyst.
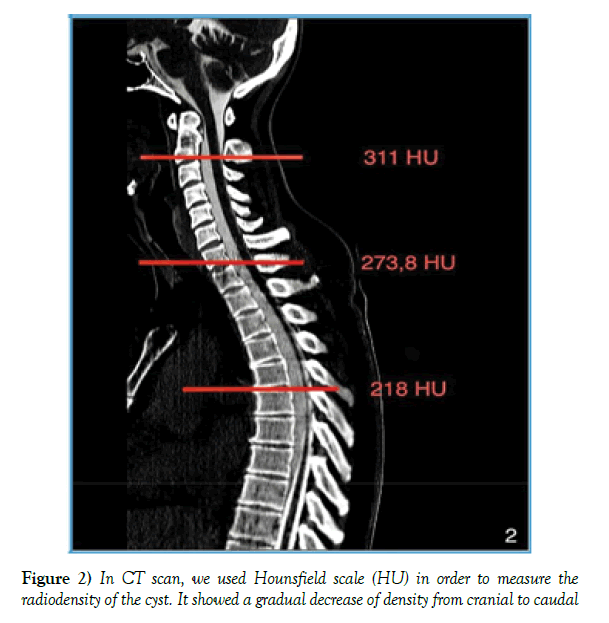
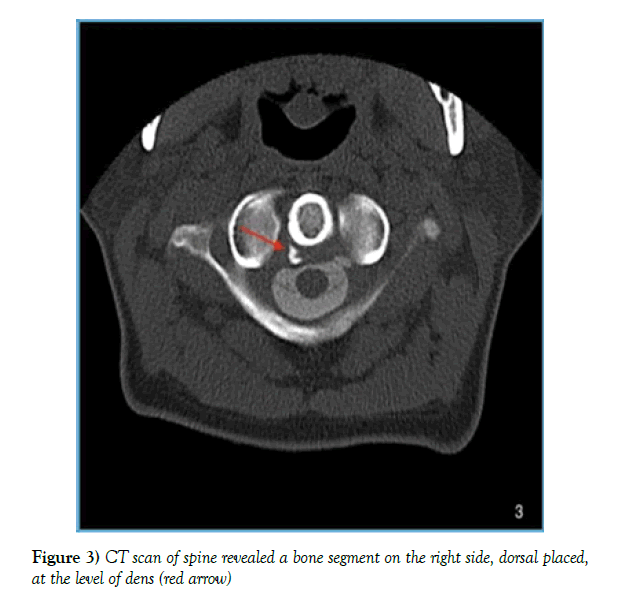
Prevedello et al. [1] described the post-myelogram CT performed with a latency of 3 hours, and both the cyst and the dural sac were contrasted and no fistulous origin could be identified. We opted for an immediate post-puncture implentation of the post -myelogram CT to take into account the cerebrospinal fluid dynamics and possibly show an early contrasting of the existing fistula point. The CT showed a connection with the same density as the dural sac at the level of C2/3 on the left, with a distinct density gradient in the anterior cyst extended from C2 to Th8/9 (Figure 2). Furthermore, at the level of the dens axis dorsolateral on the right, there is a round bony fragment (Figure 3).
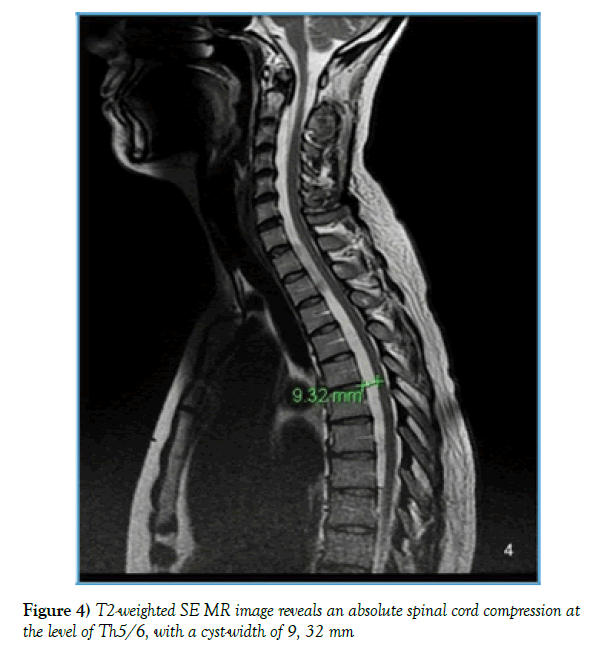
The complementary MRI (3 Tesla Siemens Magnetom skyra) showed an intraspinal extradural cystic lesion hypo-intense in T1W and hyper- intense in T2W with an extension of C2/3 to Th8/9 with a maximum diameter of 9, 32 mm at the level Th5/6, with a relevant compression of the dural sac with long-range stenosis, without myelopathy signal (Figure 4).
Figure 4) T2-weighted SE MR image reveals an absolute spinal cord compression at the level of Th5/6, with a cyst-width of 9, 32 mm
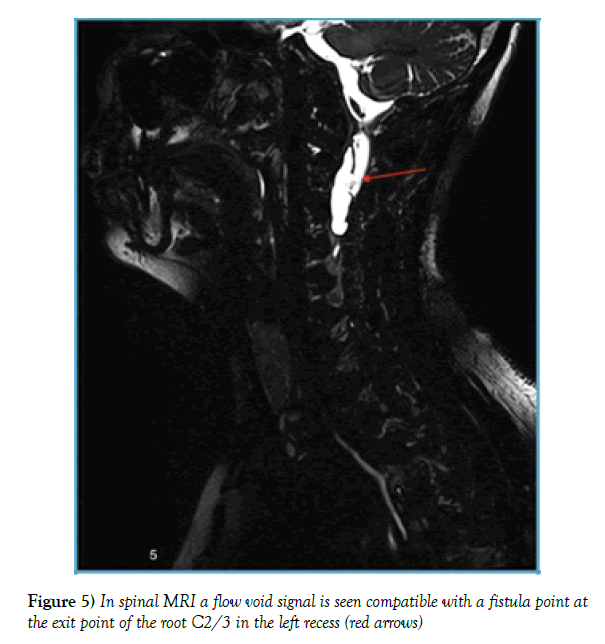
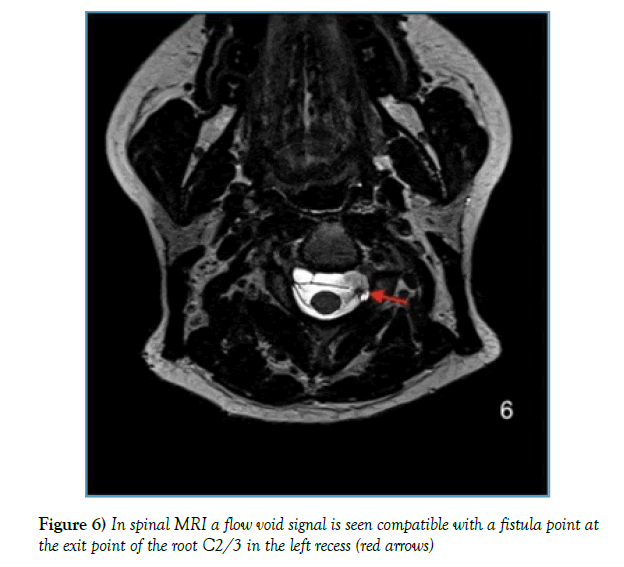
Corresponding to the post-myelogram CT, a flow void signal is seen compatible with a fistula point at the exit point of the root C2/3 in the left recess (Figures 5 and 6).
Operation
After reviewing all radiological findings, we decided to perform a duraplasty in order in order to repair the ventral dural defect at the level of C2 left-sided, following by an surgical decompression of spinal cord at the level with the major compression Th5-6, so as to relieve the compression of the cord. We performed a middle line posterior approach in semi-sitting position at the level of upper cervical spine. Under Fluoroscopy we approached the inferior surface of lamina C2, which was drilled out, so that a hemi-laminectomy was achieved. Directly under the ligamentum flavum we saw the intact dural dorsal wall. It was opened vertically at that level, following removal of CSF. Lower we saw the spinal cord displaced on the right side and the C3 left nerve root. Medial and cranial of C3- root there was a 2 mm dural defect. That defect was plugged with an autologous subcutaneous fat graft and collagen sponge (Tachosil). At the end, we performed a duraplasty and laminoplasty of C2. (Fixation of lamina with screws).
Subsequently a microsurgical Th5-6 fenestration in prone position and an hemi-laminectomy right-sided was performed, in order to access the epidural space. Directly under the ligamentum flavum we identified a tense cystic wall, which was in close connection with dural wall. The dorsal cyst wall could be identified and carefully dissected from the thecal sac. The cyst wall was opened following removal of CSF-like fluid, and the cyst was immediately decompressed. The thecal sac was completely relaxed and exhibited a normal pulsation. Neither dural defect or any CSF leakage could be observed. At the end, a hemostasis of paravertebral venous plexous was performed and the wound was closed.
Histopathological findings
Pseudocyst. No signs of arachnoidal cells in material.
Postoperative course
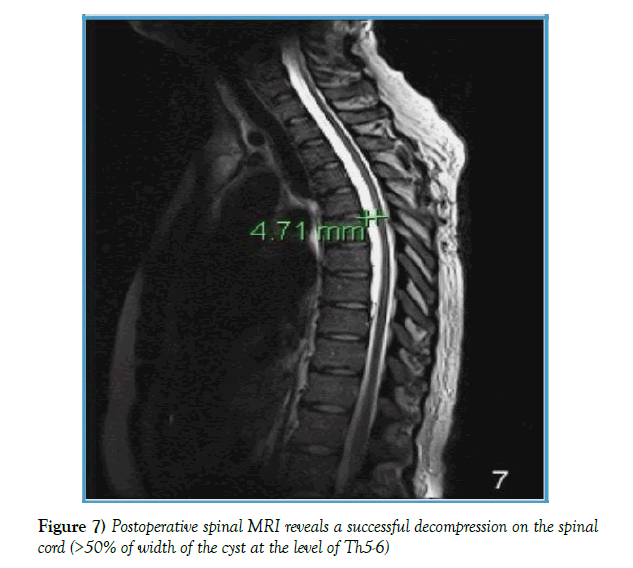
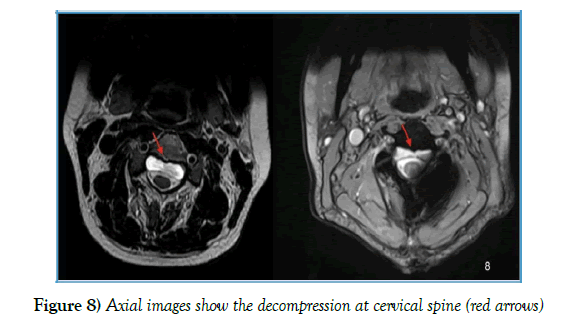
The patient stayed 7 days in a regular ward, where she was mobilized. The patient’s hypoesthesia improved direct postoperatively. Dysmetria improved markedly. By the time of discharge there was still a light hypoesthesia of the lateral part of the left foot, as well as a bradykinesia, with slight slowed movement by point - to-point test on the left. Follow- up MRI scan showed the successful decompression of spinal cord (4, 71 mm from 9, 32 mm), as well as the repair of dural defect (Figures 7 and 8). In clinical and radiological follow- up after 3 months showed an unremarkable neurological status, as well as an unchanged MRI.
Discussion
Background and clinical symptomatology
Spinal pseudocysts are rare extradural lesions, collections of CSF, which could be result of a damage of dural- arachnoidal layer. There is still an inconsistent of nomenclature in literature, as they could be mentioned as meningeal pseudocyst, extradural cysts, arachnoid cysts, arachnoidal diverticulum, lumbar cysts, spurious meningocele, acquired meningocele, iatrogenic meningo- cele [2,3]. They could be classified into 3 categories: congenital, traumatic and iatrogenic. The majority of pseudomeningoceles are iatrogenic after incidental durotomies during spinal surgery [2,4]. The true incidence of pseudomeningoceles is still unknown, as many of them still remain asymptomatic. In principle, they could be asymptomatic for years, till the time, when that mass enlarges and compress the neighboring structures, even more the spinal cord (progressive myelopathy) [5]. Depending on the level of the lesion, such a cyst could be presented as: extremities weakness, low-back-pain, radicular pain, nausea or vomiting, headaches or meningismus signs, delayed myelopathy [2,6-9]. The median interval between onset of symptoms and surgery was about 9 months, ranging from 1- 96 months [6].
Pathogenesis
There is a lack of information’s about precise mechanism of pathogenesis of spinal pseudocyst, but the cause seems to be a damage of the dural-arachnoidal layer. It may occur due to an operation (e.x Laminotomie, incidental durotomy), due to a trauma, or even a congenital abnormality (dural malformations). Subsequently the formation of the cyst seems to be a mechanical process, which depends on the size of dural-arachnoidal defect, pressure of spinal fluid, and resistance of the surrounding soft tissues. There have been proposed some pathogenic mechanism such as: 1) Check- valve mechanism, 2) Hyperosmolar collection of fluid within the cyst, 3) Secretion of fluid by cells lining the cyst wall [5,9]. The theory that CSF leak out via a dural defect and the rootlet caught in the defect interrupts its return into the subarachnoid space (valve-mechanism) seems to me the most widely accepted [5,9].
Treatment options
It’s necessary to determine the indications for surgical treatment of spinal cysts. In general, small cysts in asymptomatic patients could be conservatively treated, with observation, and only if a progressive neurological dysfunction appears, might an operation be considered. [5,9,10]. In order to determine the appropriate surgical treatment, surgeons have to take into consideration an important parameter. That’s the communication of the cyst with subarachnoid space [5,9]. In cysts without any communicating tract, a complete surgical excision has to be the goal. On the other side in those cysts with a communicating tract, first goal remains the watertight repair of the dural defect, so as to eradicate the ball-valve mechanism [5,9,10].
In literature there were also some authors, who propose a cyst- toperitoneal shunting when the dural defect is large and difficult to be repaired, but it should be the last option [5]. In cases in which the cyst is long extended or adherent to spinal canal, it could be performed the technique of marsupialization of the cyst by resecting the dorsal wall of the cyst and closing the dural defect [5,9]. Marsupialization has been reported to achieve relief from symptoms successfully [9]. In conclusion, according to Novak et al. a simple evacuation or aspiration of the cyst will result in only temporary improvement of symptoms [10].
Imaging findings and differential diagnosis
Radiological examination plays an important role in the diagnosis of an intraspinal pseudocyst. Plain X-rays are not so useful, as there provide only some indirect signs about the cyst, such as wall effects, enlarged spinal canal, bony erosions of the spine [9]. To set the diagnosis a MRI -scan and a adjunctive computerized tomography with myelography have to be performed [9,11]. MRI has great sensitivity and specificity for CSF-containing lesions.
In differential diagnosis, there are all these intraspinal cystic lesion, which have to taken into account. Specifically, about intraspinal extradural lesions, these could be synovial cysts, ganglion cysts, Tarlov’s peri- neural cysts, extradural arachnoid cysts, dermoid cysts, and neuromas with cystic changes [3,12,13]. Goyal et al. [3] observed that extradural arach- noid cysts were synonymous with sacral meningoceles, arachnoid pouches, arachnoid diverticula and meningeal cysts. Nabors et al. have classified the spinal meningeal cysts into three major categories: extradural cysts with nerve root fibers (Type-I/extradural aranoid cyst); extradural cyst without nerve root fibers (Type-II/sacral meningocele); and intradural cysts (Type- III).
Conclusion
Spinal cord decompression for extending lesions in symptomatic patients, and if it’s possible with a total removal of cyst, as well as repair of the dural defect is the primary treatment for spinal extradural pseudocyst.
REFERENCES
- Prevedello, Daniel MS, Andrei K, et al. Ventral extradural spinal meningeal cyst causing cord compression: neurosurgical treatment Arquivos de Neuro-Psiquiatria. 2005;63:855-8.
- Narverud SF, Chandran H, Dharmendra G, et al. Calcified pseudomeningocele of the lumbar spine: a review. Eur Spine J. 2012;22:443-9.
- Goyal RN, Benoit BG, Belanger JM, et al. Intraspinal cysts: a classification and literature review. Spine. 1987;12(3):209-13.
- Couture D, Branch CL Jr. Spinal pseudomeningoceles and cerebrospinal fluid fistulas. Neurosurg Focus. 2003;15(6):E6.
- Liu JK, Peter K, Schmidt MH, et al. Spinal extradural arachnoid cysts: clinical, radiological, and surgical features. Neurosurg Focus. 2007;22(2):1-5.
- Garg K, Kale SS, Sharma BS, et al. Spinal arachnoid cysts – our experience and review of literature. Br J Neurosurg. 2017;31(2):172-8
- Serge M, Marlene A, Seiler RW, et al. Multiple spinal extradural meningeal cysts presenting as acute paraplegia Case report and review of the literature. J Neurosurg Spine. 2007;6:465-72.
- Cho HY, Lee SH, Kim ES, et al. Symptomatic Large Spinal Extradural Arachnoid Cyst: A Case Report. Korean J Spine. 2015;12(3):217-20.
- Choi JY KS, Lee WS, Sung KH. Spinal extradural arach- noid cyst. Acta Neurochir (Wien). 2006;148:579-85.
- Novak L, Dobai J, Nemeth T, et al. Spinal extradural arachnoid cyst causing cord compression in a 15-year-old girl: a case report. Zentralblatt für Neurochirurgie. 2005;66(1):43-6.
- Rimmelin A, Clouet PL, Salatino S, et al. Imaging of thoracic and lumbar spinal extradural arachnoid cysts: report of two cases. Neuroradiology. 1997;39(3):203–6.
- Kono K, Nakamura H, Inoue Y, et al. Intraspinal Extradural Cysts Communicating with Adjacent Herniated Disks: Imaging Characteristics and Possible Pathogenesis. AJNR Am J Neuroradiol. 1999;20(7):1373-7.
- Nabors MW, Pait TG, Byrd EB, et al. Updated assessment and current classification of spinal meningeal cysts. J Neurosurg. 1988;68(3):366-377.