Meningocele of petrous apex: A case report, literature review of differential diagnosis and its radiological findings
2 Department of Radiology, Clemens Hospital, Academic Hospital of Münster University, Münster, Germany
Received: 17-Jan-2018 Accepted Date: Feb 03, 2018; Published: 10-Feb-2018
Citation: Fistouris P, Winkelmann D, Wessling J, et al. Meningocele of petrous apex: A case report, literature review of differential diagnosis and its radiological findings. J Neurol Clin Neurosci. 2018;2(1):24-26.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Meningoceles consist of meninges and CSF that have herniated through a skull defect, usually located in the midline. They are seen more commonly in children and very rarely in adults. We report a case of a 65-year-old patient, who was diagnosed with a osteolytic lesion in the right petrous apex, after computed tomography of the head was performed. A magnetic resonance imaging scan of the brain showed a cystic CSF collection through the defect. Below we present our experience of a meningocele of petrous apex, and by the way we provide a brief review of its differential diagnosis, as well as its relevant imaging findings.
Keywords
Meningocele; Petrous apex lesions; Differential diagnosis; Imaging findings
Introduction
Meningoceles are protrusions of the meninges through points of weakness, either in spinal cord or intracranial. They are usually connected with NTDs (neural tube defects) and spina bifida in children. Intracranial meningoceles on the other side seem to be rare lesions, which depending on their size and location could provoke various clinical manifestations. Mainly they present with CSF leak and meningitis, headaches or more rarely seizures. Their differential diagnosis includes a wide variety of lesions, and on that purpose radiological findings seem to play the essential role. Below we present our case report and we provide a brief review of the corresponding differential diagnosis and its imaging findings.
Case Report
A 65-year-old female patient presented in our clinics after a cranial ct-scan, which showed an osteolytic lesion located in the right petrous apex. The patient was complaining about repeated falls during the previous few months. Furthermore she had been suffering from occasional headaches during the previous 15 years [1]. Intact neurological status. The cranial ct-scan showed a lytic lesion in the skull, as an extensive nonaggressive erosion of right petrous apex. Given these findings, we performed an MRI examination of the brain, which showed a lesion with high signal intensivity on T2- weighed magnetic resonance imaging (MRI), which is in contact with Meckel’s cave. A radiologic diagnosis of an meningocele was made. Furthermore an adjacement to the ICA (internal carotid artery) was identified [2-5]. The patient underwent a transnasal / transphenoidal operation. The meningocele was reduced and the defect was plugged with an autologous abdominal fat graft and fibrin glue. Transnasal-transphenoidal approach is a safe, effective, minimally invasive and well tolerated approach for these lesions, allowing to resect the herniated tissue and repair the dural and bone defect [6,7]. Postoperative imaging revealed fat filling the petrous apex up to the osseous defect and mild pneumatocephalus. A small amount of tissue was sent to pathology for intraoperative diagnosis [8]. The histological diagnose was bone and connective tissue; no adjuvant therapy was administered.
Discussion
Meningoceles are classified in this group of malformations of craniofacial skeleton, which simply represent defects in scalp, bone, meninges or brain [9-11]. They consist a subtype of encephaloceles, as their calvarian defect is associated only with herniation of leptomeninges and cerebrospinal fluid (CSF). In literature series, its shown variable incidences of different types of cephaloceles among patients from different geographic locations, race and sex, as well as different associations with neural tube defects. Meningoceles are most commonly congenital in origin and have an estimated incidence of one in every 35,000 live births. Spontaneous meningoceles or meningoencephaloceles account for only 8.6% of all meningoencephaloceles [12]. The relative incidences of cranial meningoceles versus meningoencephaloceles vary widely, partly as a function of the type of cephalocele. In literature series, Simpson [13,14] found that 24% of reported cephaloceles were cranial meningoceles and 76% meningoencephaloceles.
Etiology
Meningoceles and therefore encephaloceles can be divided according to etiology into:
1. Congenital (the most common)
2. Secondary (traumatic, postsurgical, neoplastic, inflammatory- history of chronic otitis media, or even with idiopathic intracranial hypertension syndrome)
3. Spontaneous (rare)
They may present either as a single isolated congenital anomaly or smaller number of cephaloceles as variable parts of known syndromes [1]. Encephaloceles-meningoceles are most commonly associated with Meckel’s syndrome and amniotic band syndrome [11]. Congenital encephaloceles-meningoceles are divided into three major types: cranial vault, frontoethmoidal, and basal. Secondary encephaloceles-menigoceles are acquired lesions that are most commonly the result of trauma or postsurgical defects. According Bialer et al. meningoceles are significantly more common with patients with idiopathic intracranial hypertension (IIH) [12].
Clinical signs
The petrous apex is a 4-sided-pyramid with its apex anteromedial and its base posterolateral. It is wedged in at the base of skull, anteromedial to the inner ear, between the the posterior border of the great wing of the sphenoid bone and the basilar part of occipital bone. The lesions of petrous apex can affect the surrounding anatomical structures (such as ICA, trigeminal ganglion, sixth cranial nerve, jugular bulb, inferior and superior petrosal sinuses). Thats the reason why these lesions can be presented with various symptoms and clinical signs, which are dependent on the nature of pathologic process, size and location. Muckle and colleagues [3] reported that the most common presenting symptoms were hearing loss, dizziness, headaches, and tinnitus. Decreased cranial nerve V function was present in 22%. Other symptoms can be sixth cranial nerve palsy and diplopia. Symptoms often present months or years before diagnosis, and incidental discovery is not uncommon.
Diagnosis
Diagnosis and management of petrous apex lesion present unique challenges secondary to their centralized location and their anatomical adjacements. Imaging studies (CT-Scan, MRI) are the primary method used to diagnose these lesions. Subsequently we report a differential diagnosis of petrous apex lesions, as well as their characteristics on imaging findings.
Differential diagnosis
Generally petrous apex lesions can be dividen into 4 categories:
1. Inflammatory / Cystic
2. Cerebrospinal fluid / Cephaloceles
3. Neoplasms (such as Chordosarcoma, Chordoma, meningeoma, metastasis)
4. Other rare lesions (Schwanommas, Paragaggliomas)
More specially and regarding cephaloceles-meningoceles we have to take into consideration the non-neoplasmatic lesions. Thy are representing an amount of more than 90% of all petrous apex lesions. Depending on pathophysiology, the majority of nonneoplastic petrous apex lesions are inflammatory complications of air cell diesease. Pneumatization of petrous apex occurs when epithelium-lined air cells develop as medial communication from the mastoid air cells. The air cells of the petrous apex are susceptible to similar pathologic processes that occur in the mastoid segment including obstruction, opacification, inflammation, and infection.
We report a review of differential diagnosis of these lesions (Table 1).
| Petrous apex lesion Inflammatory/Cystic |
Characteristics/Pathophysiology |
|---|---|
| Cholesterin granulom/Xanthogranulom | Cholesterin crystal, Foam cells. Anaerobic catabolism of blood and blood products. |
| Mucocele Cholosteatoma/Epidermoid |
Obstructed mucosalized air space. |
| Epithelial cell remnants/multipotential embryonic cells. | |
| Effusion or retained | Infection of middle ear or mastoid -> trapping of residual fluid in the petrous apex. |
| Cerebrospinal fluid cysts and cephaloceles | |
| Meningocele/CSF cyst/arachnoid cyst | Points of weakness, splitting of meninges, skull base. |
| Cephaloceles/Meckel’s cave diverticulum | Protrusion of arachnoid or dura from Meckel’s cave. |
| Abszess/petrous apicis | Spread of infection through preformed air-cell tracts in temporal bone/usual otitis media. |
| Aneurysma | Petrous carotis interna aneurysma/posttraumatic, congenital, mycotic. |
| Asymmetric pneumatization | Asymmetrie of air cells (5-10 % of all individualls). 10-30 % air cells in temporal bones. |
Table 1: Differential diagnosis of these lesions
Imaging findings
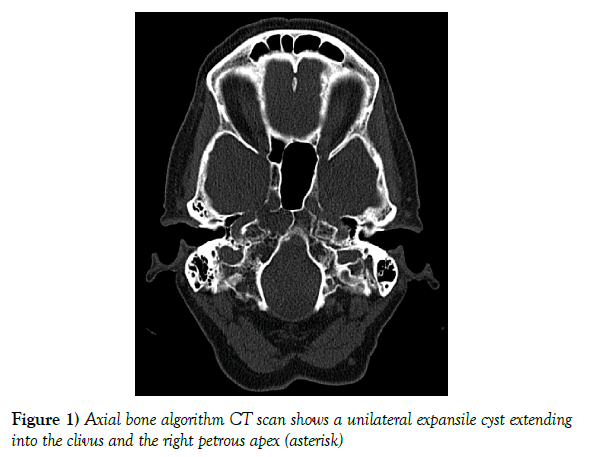
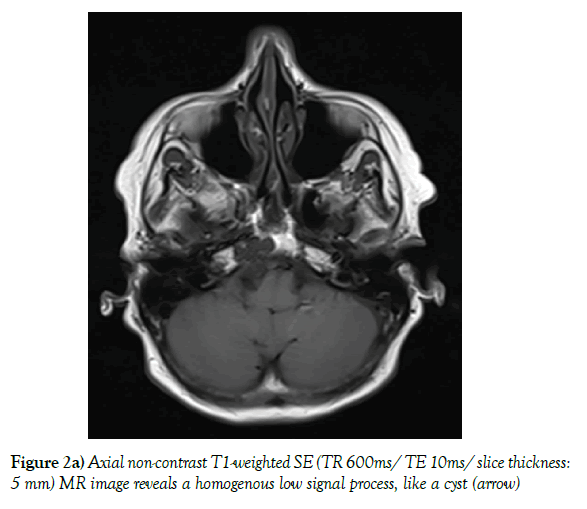
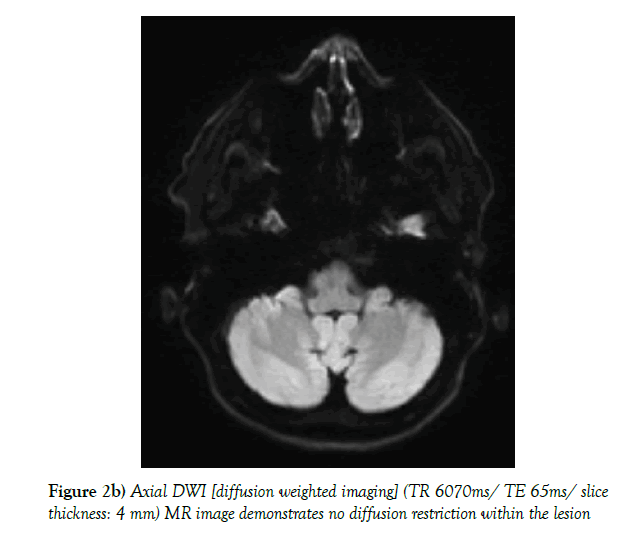
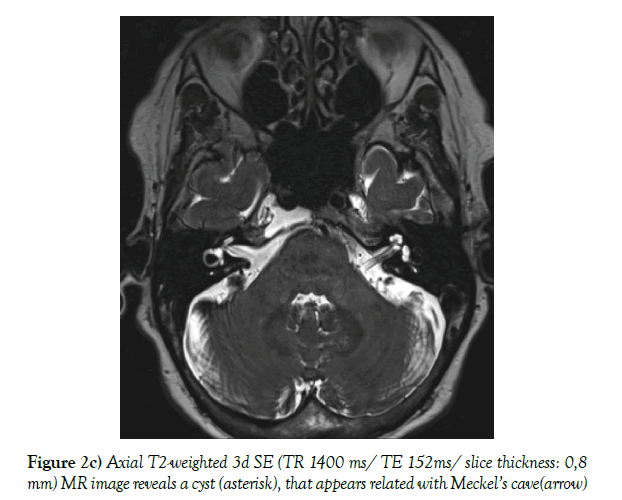
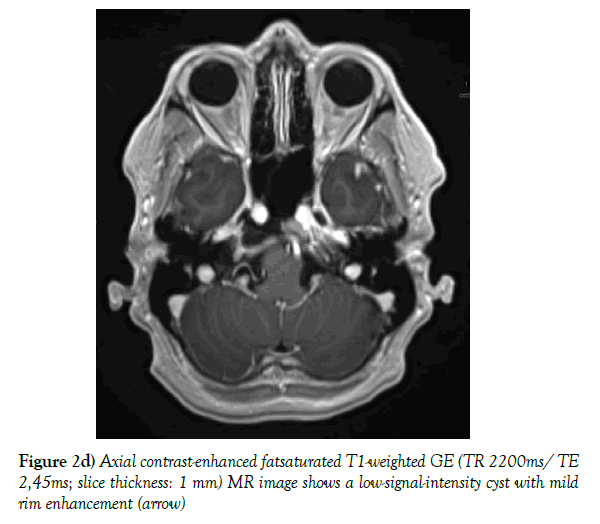
A low-dose non-contrast CT scan (80 mA & 120 kV) was performed with a Toshiba Aquilion CX CT helical scanner with wide window width (2700 HU) and level (400 HU) using a bone filter kernel. MR imaging was performed on a Siemens Skyra 3-Tesla MR unit with standard T1-weighted (spin echo; TR 600 ms/TE 20 ms; 5 mm slice thickness), T2-weigted 3d (spin echo; TR 1400 ms/ TE 152 ms; 0,8 mm slice thickness) and T2-weighted inversion recovery (spin-echo; TR 9000 ms/ TE 81 ms; 5 mm slice thickness) sequences; additional diffusion weighted imaging (RESOLVE DWI; TR 6070 ms/ TE 65 ms, b=0 & 1000; 4 mm slice thickness), and a contrast-enhanced T1 weighted 3d (gradient echo [MP RAGE]; TR 2200 ms/ TE 2,45 ms; 1 mm slice thickness) sequence was executed. The CT scan shows a unilateral expansile cyst with a maximum diameter of 21 mm and thinned sclerotic but sharply defined osseous margins in the right petrous apex, the body of the sphenoid bone and the clivus (Figure 1). The lesion erodes the bone to the posterior portion of Meckel’s cave, the carotid canal wall and the petrooccipital fissure. The cyst content has got a homogenous fluid-like attenuation (10-20 HU). T1-weighted MR images show a homogenous low signal, non-enhancing cyst content and a thin wall of low signal intensity encircling the cyst, with mild rim enhancement. T2-weighted MR images reveal fluid-like homogeneous hyperintensity. There is no diffusion restriction within the cyst. The cyst neither extends into the cerebellopontine angle nor into the inner or middle ear structures. In the contrast enhanced MR images and the 3d T2-weighted images the cyst appears related with Meckel’s cave (Figure 2a-d).
Figure 2b) Axial DWI [diffusion weighted imaging] (TR 6070ms/ TE 65ms/ slice thickness: 4 mm) MR image demonstrates no diffusion restriction within the lesion
REFERENCES
- Naidich TP, Altman NR, Braffman BH, et al. Cephaloceles and re- lated malformations. AJNR Am J Neuroradiol. 1992;13(2):655-90.
- Moore KR, Harnsberger HR, Shelton C, et al. ‘Leave me alone’ le- sions of the petrous apex. AJNR Am J Neuroradiol.1998;19(4):733-8.
- Muckle RP, Cruz A, Lo WM. Petrous apex lesions. Am J Otol. 1998;19(2):219-25.
- Isaacson B, Kutz JW, Roland PS. Lesions of Petrous Apex : Diagnosis and Management. Otolaryngol Clin N Am. 2007;40(3):479-519.
- Kevin RM, Nancy JF, Harnsberger HR, et al. Petrous Apex Cephaloceles. AJNR Am J Neuroradiol. 2001;22:1867-71.
- Çavusoglu M, Duran S, Hatipoglu HG, et al. Petrous apex cephalocoele: contribution of coexisting intracranial pathologies to the aetiopathogenesis. Br J Radiol. 2015;88(1048):20140721.
- Perez JA, Juncos JMM. Parietal intradiploic encephalocele: Report of a case and review of the literature. Neuroradiol J. 2015;28(3):264-7.
- Alorainy IA. Petrous apex cephalocele and empty sella: Is there any relation. Eur J Radiol. 2007;62:378-84.
- Chapman PR, Shah R, Curé JK, et al. Petrous Apex Lesions: Pictorial Review. Am J Roentgenol. 2011;196:WS26-37.
- Kou YF, Allen KP, Isaacson B. Recurrent meningitis secondary to a petrous apex meningocele. Am J Otolaryngol. 2014;35(3):405-7.
- Cohen MM, Lemire RJ. Syndromes with cephaloceles. Teratology. 1982;25(2):161-72.
- Bialer OY, Rueda MP, Bruce BB, et al. Meningoceles in idiopathic intracranial hypertension. AJR Am J Roentgenol. 2014;202(3):608-13.
- Jimenez DF, Barone CM. Encephaloceles, Meningoceles and Dermal Sinuses’ Operative Techniques in Pediatric. Neurosurgery. 2001.
- Simpson DA, David OJ, White J. Cephaloceles: treatment, outcome and antenatal diagnosis. Neurosurgery. 1984;15(1):14-21.