Behavioral disorders like psychomotor agitation and generalized tonicoclonic seizures revealing a neurocysticercosis: About an observation
2 Department of Neurosurgery, University of Zimbabwe, Harare, Zimbabwe, Email: kkabulo@gmail.com
3 Neuropsychiatric Center, Joseph Guislain of the Brothers of Charity, Republic of the Congo
4 Department of Preventive Medecine and Public Health, Faculty of Medecine, Pharmacy and Odontostomatology, University of Cheikh Anta Diop, Dakar, Senegal
Received: 23-Nov-2017 Accepted Date: Nov 27, 2017; Published: 04-Dec-2017
Citation: Ntenga P, Kabulo K, Mourabit S, et al. Behavioral disorders like psychomotor agitation and generalized tonicoclonic seizures revealing a neurocysticercosis: About an observation. J Neurol Clin Neurosci. 2017;2(1):5-7.
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Cysticercosis is an endemic disease in many developing countries. This diagnosis is based on the cystic aspect of the lesions, particularly in case of stay in endemic areas and serology. The clinical manifestations are of very variable expression and go from the completely asymptomatic form to very severe tables putting at stake the vital prognosis of the patient. We report in this article the case of a 43-year-old woman who was admitted to the neuropsychiatric center Dr. Joseph Guislain of ‘’Brothers of Charity’’ in Lubumbashi/DRC for a neurocysticercosis revealed by a behavioral disorder and generalized tonic clonic seizures. She was treated with albendazole 15 mg/kg/day/15 days, phenobarbital 100 mg/day at 20 h, haldol 5 mg/day/10 days and prednisolone 1 mg/kg/day/15 days. Its evolution was marked by a clear improvement of its clinical condition under treatment.
Keywords
Behavioral disorder; Generalized tonic-clonic seizures; Neurocysticercosis
Introduction
Cysticercosis is an endemic disease in many developing countries [1], and is currently increasing in developed countries [2]. Cysticercosis is secondary to the ingestion of cysticercus larvae, either by exogenous contamination (contaminated vegetables or rawness within the scope of the fecal peril), or by endogenous contamination by auto-infestation in a patient carrying T. solium (soiled hand brought to the mouth, digestion of rings raised in the stomach by anti-peristalsis [3].
This pathology is a helminthosis that is present in well-defined geographic areas of some countries, most of them in the process of development [4]. In some tropical countries, cerebral cysticercosis is one of the leading causes of hospitalization in neurology departments [5]. It represents, in Mexico, Madagascar or Reunion Island, the first cause of epilepsy. On the other hand, taeniasis due to T. solium, and even more so cysticercosis, has practically disappeared from Europe (with the exception of certain countries such as Portugal). The majority of cases of neurocysticercosis observed in industrialized countries is therefore imported cases, observed among immigrants from highly endemic areas and is still endemic and constitutes a public health problem in the tropics [6].
The most frequent locations of the disease are the central nervous system, striated muscles and orbit [7,8]. The diagnosis of cysticercosis is based on the cystic appearance of the lesions, particularly in case of stay in the endemic zone and on the serology [9].
Clinical symptomatology depends on the number of larvae ingested, their location and the immune response of the host [10].
We report in this article the case of a 43-year-old patient who was admitted to neuropsychiatric center Joseph Guislain of the Brothers of Charity in Lubumbashi/DRC for a neurocysticercosis revealed by a psychomotor agitation and epilepsy behavioral disorder. This case was published with the consent of the patient.
Medical Observation
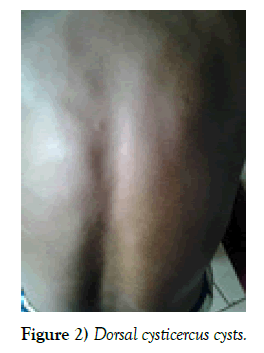
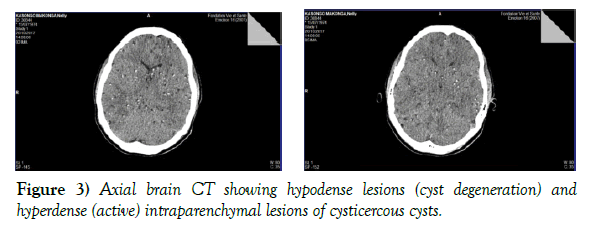
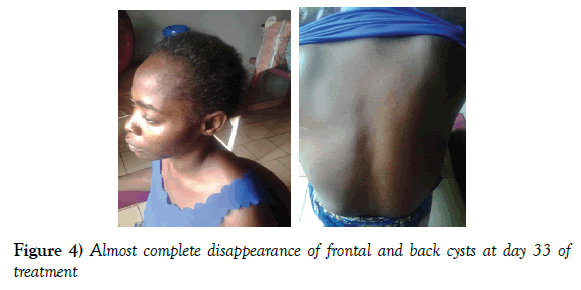
It was about Mrs KM, 43 years old, married, Congolese and resident in Lubumbashi who had been received at the neuropsychiatric center doctor Ghislain brother of the charity of Lubumbashi dated 31/10/2017 for a change of behavior made of agitation, verbal and physical aggressiveness, collectionism of clothes, logorrhea, sphincteric abandonment preceded by generalized tonic-clonic convulsive seizures. His story revealed the notion of generalized tonicoclionic convulsive seizures and moderate diffuse headaches calmed by paracetamol a year before. For her seizures, she was put under phenobarbital at 100 mg a day at night in the same center. It has been noted that she has lived in an area with intensive pig farming and consumption of pig meat. The clinical examination showed a moderate state of agitation, no signs of focusing or signs of intracranial hypertension. We noted the presence of cystic masses subcutaneous, painless, 1 to 2 cm in diameter moving under the skin in frontal (Figure 1) and dorsal (Figure 2), the ophthalmologic examination was without particularity. In front of this table a neurocysticercosis was suspected which motivated the realization of a brain scanner (Figure 3) which showed multiple hyper and hypodenseintraparenchymatous nodular lesions without mass effect. The serology (Western Blot) was requested but not performed, the other routine biological tests were normal (no leukocytosis and normal C-reactive protein), An electroencephalogram (EEG) performed under anticonvulsant treatment was normal. Thus, a neurocysticercosis was retained according to the criteria of Del Brutto et al. [11]. Treatment with oral albendazole (15 mg/kg/day) for a duration of four weeks, associated with the first seven days of corticosteroid therapy (methylprednisolone: 1 mg/kg/day), anticonvulsant (phenobartital 100 mg/day on evening at 20:00) as well as haldol tablet 5 mg/day that was introduced. In a few days, the condition of the patient improved very much compared to the initial table. The patient remained asymptomatic and was able to return home at day 16 after her hospitalization. The control scanner could not be performed due to the low financial conditions (Figure 4).
Before treatment
Figure 3) Axial brain CT showing hypodense lesions (cyst degeneration) and hyperdense (active) intraparenchymal lesions of cysticercous cysts.
After treatment
Discussion
It is described in the literature that the central nervous system is affected in cases of cysticercosis in 60 to 90% of cases, and that the period of onset of symptomatology would vary from a few months to several tens of years, even if it is often impossible to accurately determine the timing of contamination [12]. It is difficult to determine its prevalence in our environment for lack of long series.
This pathology affects people of all ages as described by Takayanagui OM et al. [13] with peak incidence in middle-aged adults. The age of our patient corroborates this statement. This same author had to show that the clinical manifestations were of very variable expression and went from completely asymptomatic form to very severe tables which could compromise the vital prognosis of the person who suffers from it. The other authors [1,14] believe that these manifestations would be a function of the number, size, location of the lesions, the evolutionary stage of the parasite, the age of the host and the variability of the immune response triggered.
Our patient had headache, tonic-clonic seizures and psychiatric agitation. This is partly in line with the study by Sciutto E et al. who showed that several clinical pictures would be described but dominated by four suggestive symptoms, particularly recent epileptic seizures, which would be the most frequent symptom, recent and progressive debilitating headaches associated or not with intracranial hypertension syndrome, focal neurological deficits and sometimes psychiatric manifestations [15]. For Sotelo J et al. [16], epileptic seizures would occur mainly when degeneration of cysts determines an inflammatory reaction, but could also occur at any stage of larval infection, especially after calcification, and it could be of all types, generalized or partial, but in the majority of cases these would be generalized crises. For Cuetter AC et al. [17], cysts located in the cerebral parenchyma are thought to be responsible for epileptic seizures in 70% of cases.
For their part, Sotelo J et al. [16], showed in their study that intracranial hypertension was more common in extraparenchymal forms, and could be accompanied by hydrocephalus, which can be acute or chronic. This intracranial hypertension would be related to a mass effect depending on the size, number of cysts, peripheral edema caused by cyst degeneration or occlusion of CSF resorption pathways by cysts with ventricular, ependymal or subcutaneous localization. -arachnoïdienne. On the other hand for Forlenza OV et al. [18], in their study they reported that focal signs would often be related to the number, size and location of parasites in patients. They reported in the same study that even if the signs reflecting localization at the level of the pyramidal system are predominant, all the focal signs could be described (hemiplegia, sensory hemispyndrome, oculomotor palsy, unilateral abnormal movements.
Forlenza OV et al. [18], described depression-type psychiatric manifestations or psychoses and that these manifestations appear to be independent of the number and type of brain lesions. Our patient was admitted in a context of psychomotor excitement, this could sufficiently show the possibility of finding the diversity of psychiatric charts in case of neurocysticercosis.
Our patient had no focal sign or sign indicating intracranial hypertension. We believe this would be related to the size of the intraparenchymal cysts and the absence of cysts that can disrupt cerebrospinal fluid dynamics. However, a medium and long-term follow-up is necessary in this one. Coulibaly B et al. [19], reported in their observation in a 70-year-old patient the presence of gait disturbances, idieational slowing and paresis of the right hemi-body with a biological assessment that was normal.
It is known that neurocysticercosis is a leading cause of acquired epilepsy in developing countries [1], and that in the world the annual mortality rate related to this pathology is greater than 50,000 people [4].
Cysts located in the cerebral parenchyma are responsible for epileptic seizures in 70% of cases. Extraparenchymal cysts (subdural or ventricular) cause intracranial hypertension due to mass effects, CSF blockage, local inflammation or fibrosis. Intraventricular cysts complicated by acute hydrocephalus are fatal in 13% of cases [17].
For Mr. Boussard et al. [20], neurocysticercosis is the most common neuroparasitosis in the world and is clinically manifested by epileptic seizures and headaches. Other more severe charts, with intracranial hypertension, stroke, could put the patient’s vital prognosis at risk. Thus we think that the pathogenic power of cysticerci would be related to their location and the severity of the manifestations could depend on their number.
Cerebrospinal fluid analysis was normal in our case. Garcia HH et al. also found that cerebrospinal fluid was normal in 50% in his patients, but sometimes the presence of moderate pleocytosis, hyperproteinorachia and hypoglycorachia. The same author reported the performance of MRI with a sensitivity of 80% for the detection of intraventricular cysts in case of intracranial hypertension. On the other hand Cuetter AC et al. [17], emphasized the importance of the T1 sequence in identifying inflammation associated with degeneration of cerebral parenchymal cysts. Some other authors have demonstrated 98% sensitivity and 100% specificity of the wester blot in terms of biological diagnosis, but these values fall (less than 50% of tests are positive) and false negatives are common in case of lesion or if the lesions are calcified [21,22].
Contradictions spread in chain on the antiepileptic treatment in case of neurocysticercosis. For some only antiepileptic treatment could guarantee the patient a quality of life and comfort lasting, for others who oppose the anti-helminthic treatment affirm that the effectiveness of this treatment is only the reflect of the natural evolution of the disease, and even untreated lesions would spontaneously disappear or calcify. Conversely, other studies show that the control of epilepsies is better after anti-helminthic treatment and that an unknown epileptic patient should not receive prophylaxis made of antiepileptic therapy [1]. However, even after curative treatment, we believe that calcified larvae will remain epileptogenic and anticonvulsant treatment should be continued and may be stopped only after two years after stabilization and disappearance of seizures.
In our observation, we have stabilized epileptic seizures with phenobarbital, and therefore this contrasts the theory of epileptic seizures of neurocysticercosis that respond well to carbamazepine monotherapy, sodium valproate or phenytoin. We plan to continue with the same treatment for fear of recurrence because they are active forms. This treatment has been combined with corticosteroid therapy to prevent cerebral edema, although it is currently under discussion [23].
In his study, Garcia HH et al., was able to show the risk of recurrence despite conventional treatment with albendazole or praziquantel (15-day courses of treatment), especially in the case of multiple lesions, and proposed prolonged sequential treatment with albendazole and praziquantel, given a different sensitivity of each cyst to each drug, for the same patient [1], this joins our phylosophy.
Conclusion
Neurocysticercosis remains a real public health problem given the risks faced by the patient in resource-limited settings with a low level of hygiene and a low technical platform for explorations. To think about it, to make the diagnosis of presumption and to take the patient quickly in charge would be prudent from the practitioner.
REFERENCES
- Garcia HH, Gonzalez AE, Evans CAW, et al. Tænia soliumcysticercosis. Lancet. 2003;362:547–56.
- White Jr AC. Neurocysticercosis: updates on epidemiology, pathogenesis, diagnosis and management. Annu Rev Med. 2000;51:187–206.
- Roudier M, Maillard A, Brousse D, et al. Cysticercosis in Guadeloupe. Ann Pathol. 2000;20:238–40.
- Roman G, Sotelo J, Del Brutto OH, et al. A proposal to declare eurocysticercosis an international reportable diseases. Bull WHO. 2000;78:399-406.
- Del Brutto OH. Neurocysticercosis. Current Opin Neurology. 1997;10:268-72.
- Rajaonarison P, Ralammboson S, Andriamamonjy C, et al. Diagnostic de neurocysticercose : à propos d’un cas. ArchInst Pasteur de Madagascar. 2001;67:53-6.
- Hawk MW, Shahlaie K, Kim KD, et al. Neurocysticercosis: a review. Surg Neurol. 2005;63:123-32.
- Rath S, Honavar SG, Naik M, et al. Orbital cysticercosis: clinical manifestations, diagnosis, management, and outcome. Ophthalmology. 2010;117:600-5.
- Gekeler F, Eichenlaub S, Mendoza EG, et al. Sensitivity and specificity of ELISA and immunoblot for diagnosing neurocysticercosis. Eur J Clin Microbiol Infect Dis. 2002;21:227-9.
- Kraft R. Cysticercosis: an emerging parasitic disease. Am Fam Physician. 2007;76:91-6.
- Del Brutto OH, Rajshekhar V, White Jr AC, et al. Proposed diagnostic criteria for neurocysticercosis. Neurol. 2001;57(2):177-83.
- Flisser A. Taeniasis and cysticercosis due to Taeniasolium. Prog Clin Parasitol. 1994;4:77-116.
- Takayanagui OM, Odashima NS. Clinical aspects of neurocysticercosis. Parasitol Int. 2006;55:S111-5.
- Fleury A, Dessein A, Preux PM, et al. Symptomatic humanneurocysticercosis age, sex and exposure factors relating with disease heterogeneity. J Neurol. 2004;251(7):830-7.
- Sciutto E, Fragoso G, Fleury A, et al. Taeniasolium disease in humans and pigs: an ancient parasitosis disease rooted in developing countries and emerging as a major health problem of global dimensions. Microbes Infect. 2000;2(15):1875-90.
- Sotelo J, Marin C. Hydrocephalus secondary to cysticercoticarachnoiditis. A long-term follow-up review of 92 cases. J Neurosurg. 1987;66(5):686-9.
- Cuetter AC, Andrews RJ. Intraventricular neurocysticercosis: 18 consecutive patients and review of literature. Neurosurg Focus. 2002;12(6):e5.
- Forlenza OV, Filho AH, Nobrega JP, et al. Psychiatric manifestations of neurocysticercosis: a study of 38 patients from a neurology clinic in Brazil. J Neurol Neurosurg Psychiatry. 1997;62(6): 612-6.
- Coulibaly B, Gautier G, Fuentes S, et al. Neurocysticercose cérébrale au stade de dégénérescence :diagnostic différentiel des métastases cérébrales, revue neurologique. 2008;164:948–52.
- Boussard M, Million L, Grenouillet F, et al. Prévention ettraitementdela cysticercose. Journal des Anti-infectieux 2012;14:143-50.
- Chabasse D, Guiguen C. Parasitosesetmycoses desrégions tempéréesettropicales, Anofel. Paris: Elsevier Masson;2010.
- Almeida CR, Ojopi EP, Nunes CM, et al. Taeniasolium DNA is present in the cerebrospinal fluido neurocysticercosis patients and can be used for diagnosis. Eur Arch Psychiatry Clin Neurosci. 2006;256(5):307-10.
- Nash TE. Human case management and treatment of cysticercosis. Acta Trop. 2003;87(1):61-9.